Meet Sigma2, a New Aβ Receptor?
Quick Links
Prion protein, insulin receptors, immunoglobulin-like receptors, PirB, unspecified membrane phospholipids, the list goes on … Now, add another purported Aβ receptor. At the Society for Neuroscience annual meeting in Washington, D.C., November 15 to 19, researchers led by Susan Catalano at the Pittsburgh-based biotech Cognition Therapeutics (CogRx) reported that Aβ interacts with the sigma2 receptor, which also goes by the alias progesterone receptor membrane component 1. Small molecule ligands developed by the company bind sigma2/PGRMC1 and reportedly restore cognitive function in transgenic mouse models of AD. The ligands block all manner of Aβ species from accessing the receptor. They even displace Aβ oligomers from brain tissue of Alzheimer’s disease patients, Catalano claimed. Could sigma2 be the real McCoy? "What is different is that Aβ no longer binds when the researchers knocked down sigma2," said John Cirrito, Washington University, St. Louis. "The pharmacology was interesting, but the genetics was the clincher for me," he said. Cirrito consults for the company.
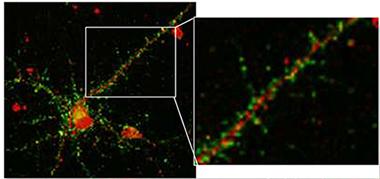
Aβ Receptor?
Primary hippocampal neurons express sigma2/PGRMC1 (red), which often co-localizes with synaptophysin (green) in synaptic boutons. [Image courtesy of Izzo et al., PLoS One.]
Iryna Benilova from K.U. Leuven, Belgium, wrote to Alzforum, "This study is outstanding in three main aspects: compound-mediated displacement of bound oligomers has not been shown for other Aβ receptors; the neutralizing effect of sigma-2 allosteric antagonists holds for a remarkably broad array of Aβ species; and finally, the in vivo effects of sigma-2 antagonists on synapses and memory in two AD mouse models point to an interesting therapeutic potential of these compounds" (see full comment below).
Despite its name, sigma2/PGRMC1 does not bind progesterone. Little is known about the protein, though it seems to stabilize a variety of other receptors on the cell membrane. Catalano and colleagues identified sigma2 after running an unbiased screen for chemicals that prevent Aβ from interfering with the transport of vesicles to the cell surface. At the SfN meeting in San Diego in 2010, Catalano introduced CT0093. Found in a proprietary chemical library, this molecule restored vesicle trafficking in primary hippocampal neurons treated with Aβ oligomers from human tissue (see Dec 2010 conference story). Since then, the company scientists have identified other compounds with similar properties, and those eventually led them to the sigma2 receptor. They described their work on five posters at this year's meeting and in two recent back-to-back papers in PLoS One (see Izzo et al., 2014, and Izzo et al., 2014).
Four of the CogRx compounds—CT0109, CT0093, CT01344, and CT01346 (structures can be found in the PLoS papers)—almost completely prevented a variety of Aβ species (including synthetic oligomers) and patient-derived oligomers from binding to primary hippocampal neurons. In her poster, Kelsie Mozzoni reported that the compounds protected vesicle trafficking when added before Aβ oligomers and restored membrane trafficking when added after. They also prevented loss of synaptic spines. In vivo, the compounds readily entered the mouse brain when given subcutaneously (CT0109, CT0093) or by mouth (CT01344, CT01346). They protected animals from Aβ-associated memory loss in acute and chronic dosing regimens. These included injecting the compounds into the hippocampi of normal mice an hour before injecting Aβ oligomers into the same location, giving the compound by mouth to 11-month-old transgenic APP mice for 42 days, and a 5½-month treatment begun when APP mice were 9 months old.
To find out how these molecules work, the researchers screened 100 known cell surface receptors and proteins. The compounds bound specifically only to sigma2/PGRMC1. Interestingly, they only restored memory in the APP mice when their concentrations in the brain were sufficient to occupy 80 percent of sigma2 binding sites. Fifty percent occupancy had no behavioral effect. "That makes us believe sigma2 is the true target [for these compounds] in vivo," said Catalano.
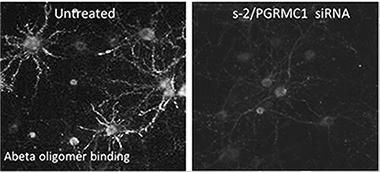
Knocking down sigma2 with siRNA eliminates Aβ binding sites. [Image courtesy of Izzo et al., PLoS One.]
Other experiments suggest these compounds are antagonists of Aβ. In her poster, Courtney Rehak showed that if she silenced the receptor in primary rat neuronal cultures using siRNA, Aβ oligomer binding plummeted more than 90 percent. Colleen Silky's work demonstrated that the CogRx compounds not only competed with Aβ for binding to primary hippocampal neurons, but displaced the peptide when they were added later. "I am not aware of any small molecule that can both prevent Aβ oligomers from binding and displace those oligomers," said Catalano. "That is a major difference with our work."
The observations extend to human tissue. Collaborating with Tara Spires-Jones, University of Edinburgh, the CogRx researchers found that CT1344 displaced endogenous Aβ from postmortem brain slices. Nicholas Izzo incubated tissue from six AD patients for one hour with either the compound or antibodies to sigma2, and saw a dose-dependent loss of Aβ immunoreactivity. In the AD brain, sigma2 expression may be dysregulated, as well. Izzo and colleagues found that brain tissue from patients with advanced dementia expressed as much of the protein as did tissue from age-matched controls, while levels of another receptor (Sigma1) had dropped by about half. The authors argue that because people with advanced dementia have lost neurons, sigma2 must be upregulated in their brain for it to match control levels.
How might sigma2 mediate Aβ toxicity? Catalano acknowledged that it is not clear if Aβ binds to sigma2, or to something else that interacts with sigma2. "Both are possibilities," she said, "The story is more subtle than just another Aβ receptor."
Since sigma2 appears to be involved in trafficking receptors on the cell surface, might it interact with some of the previously identified Aβ partners? "That's absolutely the first place you might start to look to understand the oligomer receptor complex," said Catalano. Carla Shatz, Stanford University, California, agreed. "It will be fascinating to see how their discovery meshes with our finding that PirB is a high-affinity receptor for Aβ," she wrote to Alzforum (see Sep 2013 news story).
CogRx hopes to start clinical trials next year. "In light of the recent clinical trial failures, both of our studies offer hope that there will be new (earlier) treatment avenues that do not depend on busting up plaques at times when it may be too late to have any effect," said Shatz.—Tom Fagan
References
News Citations
- San Diego: Tweakers and Tweezers—Grappling with Novel Aβ Therapies
- Immune Receptor Binds Aβ Oligomers, Spurs Synaptic Loss
Research Models Citations
Paper Citations
- Izzo NJ, Staniszewski A, To L, Fa M, Teich AF, Saeed F, Wostein H, Walko T 3rd, Vaswani A, Wardius M, Syed Z, Ravenscroft J, Mozzoni K, Silky C, Rehak C, Yurko R, Finn P, Look G, Rishton G, Safferstein H, Miller M, Johanson C, Stopa E, Windisch M, Hutter-Paier B, Shamloo M, Arancio O, LeVine H 3rd, Catalano SM. Alzheimer's therapeutics targeting amyloid beta 1-42 oligomers I: Abeta 42 oligomer binding to specific neuronal receptors is displaced by drug candidates that improve cognitive deficits. PLoS One. 2014;9(11):e111898. Epub 2014 Nov 12 PubMed.
- Izzo NJ, Xu J, Zeng C, Kirk MJ, Mozzoni K, Silky C, Rehak C, Yurko R, Look G, Rishton G, Safferstein H, Cruchaga C, Goate A, Cahill MA, Arancio O, Mach RH, Craven R, Head E, LeVine H 3rd, Spires-Jones TL, Catalano SM. Alzheimer's therapeutics targeting amyloid beta 1-42 oligomers II: Sigma-2/PGRMC1 receptors mediate Abeta 42 oligomer binding and synaptotoxicity. PLoS One. 2014;9(11):e111899. Epub 2014 Nov 12 PubMed.
Further Reading
News
- Patient-Derived Aβ Needs Prion Protein to Harm Synapses
- Keystone: Partners in Crime—Do Aβ and Prion Protein Pummel Plasticity?
- How Does Aβ Do Harm? New Clues on Insulin Signaling, Spines, Inflammation
- Aβ Downs EphB2 Kinase, Disrupts Glutamate Receptors
- Aβ Oligomers and Synaptic Dysfunction—Blame It on RAGE?
- Glutamate Receptor Links Aβ-Prion Complex with Fyn, Synaptic Damage
- Immune Cell Receptor Gives Aβ a Toxic Edge
Primary Papers
- Izzo NJ, Staniszewski A, To L, Fa M, Teich AF, Saeed F, Wostein H, Walko T 3rd, Vaswani A, Wardius M, Syed Z, Ravenscroft J, Mozzoni K, Silky C, Rehak C, Yurko R, Finn P, Look G, Rishton G, Safferstein H, Miller M, Johanson C, Stopa E, Windisch M, Hutter-Paier B, Shamloo M, Arancio O, LeVine H 3rd, Catalano SM. Alzheimer's therapeutics targeting amyloid beta 1-42 oligomers I: Abeta 42 oligomer binding to specific neuronal receptors is displaced by drug candidates that improve cognitive deficits. PLoS One. 2014;9(11):e111898. Epub 2014 Nov 12 PubMed.
- Izzo NJ, Xu J, Zeng C, Kirk MJ, Mozzoni K, Silky C, Rehak C, Yurko R, Look G, Rishton G, Safferstein H, Cruchaga C, Goate A, Cahill MA, Arancio O, Mach RH, Craven R, Head E, LeVine H 3rd, Spires-Jones TL, Catalano SM. Alzheimer's therapeutics targeting amyloid beta 1-42 oligomers II: Sigma-2/PGRMC1 receptors mediate Abeta 42 oligomer binding and synaptotoxicity. PLoS One. 2014;9(11):e111899. Epub 2014 Nov 12 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University College London
Neurons contain multiple Aβ binding sites, from membrane receptors to gangliosides. It is currently unclear whether Aβ toxicity is mediated by ligand-receptor interactions or a less-specific mechanism. Using a phenotypic assay for membrane trafficking, Izzo et al. have identified low molecular weight compounds that blocked Aβ binding to neural membranes and displaced already-bound Aβ oligomers. Of note, Aβ-induced trafficking deficits were monitored by means of the MTT assay, whereby Aβ-treated cultures displayed enhanced exocytosis of the insoluble dye (formazan). While the MTT assay is unsuitable for evaluation of Aβ-induced cell death (Wogulis et al., 2005), it is a perfect tool for the study of Aβ effects on cell membrane.
Using carefully characterized synthetic and human brain-derived Aβ aggregates, the authors demonstrate that an unprecedentedly high (more than 90 percent) level of oligomer binding is mediated by sigma-2/PGRMC1, a progesterone receptor membrane component 1 implicated in a few CNS disorders and cancer. Given the known role of sigma-2 in membrane trafficking and stabilization of transmembrane receptors, the authors raise an interesting possibility that Aβ interaction with this protein can modulate other candidate Aβ receptors as a part of a large multireceptor complex. Whether Aβ is an agonist or antagonist of sigma-2 is unclear, but caspase 3 activation, typical for sigma-2 agonists, does not seem to be involved in Aβ-induced synaptotoxic cascades. The affinity of sigma-2 for Aβ oligomers is relatively low (Kd = 0.5 μM), five to 10 times lower compared to other receptors (PrP, RAGE, and FcgR2B), and oligomers can be efficiently displaced from their binding sites by sigma-2 antagonists in a dose-dependent manner.
This study is outstanding in three main aspects: compound-mediated displacement of bound oligomers has not been shown for other Aβ receptors; the neutralizing effect of sigma-2 allosteric antagonists holds for a remarkably broad array of Aβ species; and finally, the in vivo effects of sigma-2 antagonists on synapses and memory in two AD mouse models point to the therapeutic potential of these compounds.
Make a Comment
To make a comment you must login or register.