Liver Enzyme Levitates with Exercise, Spurs Learning in Old Mice
Quick Links
It’s no longer news that exercise tones the brain as well as the body, keeping cognition sharp with age and lowering the risk of Alzheimer’s disease. Exactly how this works remains murky, however. In today’s Science, researchers led by Saul Villeda at the University of California, San Francisco, identify a liver enzyme that delivers the benefits of exercise to the brain. Once again, the findings so far are in mice.
- Gpld1 rises after exercise, suppressing complement and inflammation.
- Overexpressing Gpld1 boosts cognition in old mice.
- Is there a liver-brain axis?
The enzyme, glycosylphosphatidylinositol (GPI)-specific phospholipase D1 (Gpld1), snips GPI-linked surface proteins off cells. This alters numerous cellular processes. In particular, Gpld1 suppresses complement, part of the innate immune system. Overexpressing Gpld1 in mice stimulated their neurogenesis and sharpened their learning and memory, mimicking known effects of exercise on the brain. Moreover, plasma Gpld1 levels are higher in healthy older people who exercise than in their sedentary peers, hinting that this mouse finding could be relevant to people.
“This is an excellent paper that describes a novel axis, from liver to brain,” said Gerard Karsenty at Columbia University Medical Center in New York City. He believes the findings will open up new research avenues and suggest new candidate therapeutic molecules that may protect the brain. Karsenty was not involved in this study.
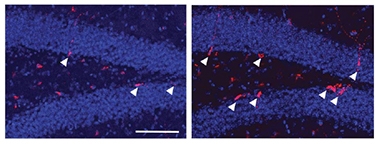
Baby Neurons. Aged mice overexpressing the enzyme Gpld1 in their livers (right) have more newborn neurons (red, arrowheads) in the hippocampus than do controls (left). Nuclei are blue. [Courtesy of Horowitz et al., Science/AAAS.]
Previous research from Tony Wyss-Coray’s lab at Stanford University first described beneficial effects of plasma from exercising mice on the brains of sedentary animals. This “runner’s plasma” was high in clusterin and other complement inhibitors, which appeared to dampen neuroinflammation (Sep 2019 news).
Villeda, who did his graduate work with Wyss-Coray, took a similar approach to find additional factors. Joint first authors Alana Horowitz and Xuelai Fan collected plasma from either 6- or 18-month-old mice that were allowed to run on a wheel, and injected that into 18-month-old sedentary mice eight times over three weeks. The shots upped BDNF in the brain by a quarter, neurogenesis by half, and also improved the old mice’s performance in the radial arm water maze and contextual fear conditioning tests.
To find the factors responsible, the authors analyzed the plasma of exercising mice by mass spectrometry. They identified 12 proteins that were consistently elevated by exercise in both age groups. They were mostly metabolic proteins made by the liver. Among the dozen, Gpld1 and serum paraoxonase 1 stood out as key. Each is involved in numerous metabolic processes, such as cholesterol efflux, hormone response, and processing ammonium, ethanolamine, and organic hydroxy compounds.

Bloodborne Tutor. Plasma from exercising mice helps sedentary mice learn better. [Courtesy of Horowitz et al., Science/AAAS.]
Overexpressing serum paraoxonase 1 in old mice did them no good. In contrast, overexpression of Gpld1 elevated the mice’s hippocampal BDNF by 40 percent and nearly tripled their neurogenesis. One to two months later, the rodents did better on the radial-arm water maze, Y maze, and object-recognition tests.
This was a surprise, since Gpld1 had not been linked previously to aging or cognition. In fact, its expression does not change with age in mice, the authors found. It goes up in the liver, but not other organs, after exercise. Its expression does not rise in the hippocampus after exercise.
How, then, might Gpld1 help the brain? Using a tagged construct, the authors found that very little of the enzyme gets past the blood-brain barrier, suggesting that it somehow exerts its effects from outside. How Gpld1 does this is still a mystery, but Villeda believes it may act by dampening peripheral inflammation, and that that may influence neuroinflammation. Supporting this, a mass spectrometry analysis of plasma from Gpld1-overexpressing mice found suppression of pro-inflammatory complement in the blood. This finding dovetails with Wyss-Coray’s data. Notably, clusterin was one of the proteins elevated in the Gpld1-overexpressing mouse plasma, suggesting it lies downstream of Gpld1 in the same pathway. “Our stories are complementary,” Villeda said.
In the brain, complement activation has been tied to synapse loss in Alzheimer’s and other disorders (Aug 2013 conference news; Nov 2015 conference news; May 2016 news). Next, Villeda will test whether complement enters the brain more readily in aged mice, and whether exercise reduces this. He will also examine whether overexpressing Gpld1 in mouse models of amyloidosis confers any benefit.
Besides suppressing complement, Gpld1 stimulated the urokinase-type plasminogen activator receptor (uPAR) signaling pathway. This pathway switches on plasminogen, which dissolves blood clots, and vitronectin, which thickens the extracellular matrix, increasing cell adhesion and clotting. In Gpld1 overexpressers, plasminogen and vitronectin protein both went down. Villeda noted that the net effect of Gpld1 on clotting is unclear. Intriguingly, plasminogen has been linked to neuroinflammation and amyloidosis (Baker et al., 2018).
Does this Gpld1-brain connection matter to people? Genetic screens have implicated uPAR as an AD risk factor, hinting at a link (Finckh et al., 2003; Wu et al., 2013). To investigate, Villeda and colleagues measured Gpld1 in plasma from 10 healthy, active seniors, finding it was about 25 percent higher than in 10 sedentary peers. The findings may further strengthen the rationale for developing exercise as a therapy in the older at-risk population (Aug 2017 news).
However, previous exercise studies suggest that participants need a moderate-intensity aerobic workout to experience cognitive benefits (Aug 2015 conference news; Aug 2015 news). Some frail elderly people are not capable of that, Villeda noted. If indeed Gpld1 mediates exercise benefits in people as it does in mice, then perhaps boosting this enzyme pharmacologically could help this population, Villeda suggested. He is trying to develop a small molecule that mimics Gpld1’s ability to cleave GPI linkages. A small molecule would make for a more tractable therapeutic than the full protein.
Most likely, a therapy to slow brain aging would combine several different plasma factors, suggested Lee Rubin at Harvard University. Rubin previously identified the growth factor GDF11 as another peripheral protein that stimulates neurogenesis without entering brain, though this one has not been linked to exercise yet (Katsimpardi et al., 2014). Villeda, Wyss-Coray, and Karsenty each found other plasma proteins associated with brain health (Gontier et al., 2018; Castellano et al., 2017; Khrimian et al., 2017). Rubin is starting a collaboration with Villeda on another project, and both sit on the advisory board of Alkahest, a biotech company in San Carlos, California, that investigates the effects of young plasma on health.
Saurabh Chatterjee at the University of South Carolina, Columbia, was particularly intrigued by the liver-brain connection. In the July 4 Journal of Neuroinflammation, Chatterjee identified a different neuroactive liver protein, lipocalin-2. Unlike Gpld1, it crosses the blood-brain barrier, and it ramps up neuroinflammation in mice. In response to lipocalin-2 entry, brain expression of BDNF drops and phosphorylated tau goes up. Chatterjee noted that the activation state of microglia affects their BDNF production, perhaps explaining this effect.
Because inflammation in the intestine, too, can have a similar detrimental effect on the brain, Chatterjee believes there may be a liver-gut-brain axis that warrants investigation. “These three organ systems may act in concert to cause neurocognitive deficits,” he suggested.
Chatterjee noted that about 200,000 veterans who served in the 1990 Gulf War suffer from chronic fatigue, cognitive deficits, and neuroinflammation due to chemical exposures. He previously linked this Gulf War illness to changes in the gut microbiome (Alhasson et al., 2017; Seth et al., 2019; Janulewicz et al., 2019). Now he plans to investigate whether Gpld1 could help reverse their deficits by administering the protein to a mouse model of Gulf War illness to evaluate its potential.—Madolyn Bowman Rogers
References
News Citations
- 'Runner Plasma' Jogs Neurogenesis, Quells Neuroinflammation in Mice
- Curbing Innate Immunity Boosts Synapses, Cognition
- Microglia Control Synapse Number in Multiple Disease States
- Microglia Prune Synapses in a Subtype of Frontotemporal Dementia
- New Dementia Trials to Test Lifestyle Interventions
- Exercise Boosts Cognition In Symptomatic Disease
- Neither Exercise Nor Supplements Boost Cognition in Two Large Studies
Paper Citations
- Baker SK, Chen ZL, Norris EH, Revenko AS, MacLeod AR, Strickland S. Blood-derived plasminogen drives brain inflammation and plaque deposition in a mouse model of Alzheimer's disease. Proc Natl Acad Sci U S A. 2018 Oct 9;115(41):E9687-E9696. Epub 2018 Sep 25 PubMed.
- Finckh U, van Hadeln K, Müller-Thomsen T, Alberici A, Binetti G, Hock C, Nitsch RM, Stoppe G, Reiss J, Gal A. Association of late-onset Alzheimer disease with a genotype of PLAU, the gene encoding urokinase-type plasminogen activator on chromosome 10q22.2. Neurogenetics. 2003 Aug;4(4):213-7. PubMed.
- Wu W, Jiang H, Wang M, Zhang D. Meta-Analysis of the Association Between Urokinase-Plasminogen Activator Gene rs2227564 Polymorphism and Alzheimer's Disease. Am J Alzheimers Dis Other Demen. 2013 Aug;28(5):517-23. PubMed.
- Katsimpardi L, Litterman NK, Schein PA, Miller CM, Loffredo FS, Wojtkiewicz GR, Chen JW, Lee RT, Wagers AJ, Rubin LL. Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science. 2014 May 9;344(6184):630-4. Epub 2014 May 5 PubMed.
- Gontier G, Iyer M, Shea JM, Bieri G, Wheatley EG, Ramalho-Santos M, Villeda SA. Tet2 Rescues Age-Related Regenerative Decline and Enhances Cognitive Function in the Adult Mouse Brain. Cell Rep. 2018 Feb 20;22(8):1974-1981. PubMed.
- Castellano JM, Mosher KI, Abbey RJ, McBride AA, James ML, Berdnik D, Shen JC, Zou B, Xie XS, Tingle M, Hinkson IV, Angst MS, Wyss-Coray T. Human umbilical cord plasma proteins revitalize hippocampal function in aged mice. Nature. 2017 Apr 19; PubMed.
- Khrimian L, Obri A, Ramos-Brossier M, Rousseaud A, Moriceau S, Nicot AS, Mera P, Kosmidis S, Karnavas T, Saudou F, Gao XB, Oury F, Kandel E, Karsenty G. Gpr158 mediates osteocalcin's regulation of cognition. J Exp Med. 2017 Oct 2;214(10):2859-2873. Epub 2017 Aug 29 PubMed.
- Alhasson F, Das S, Seth R, Dattaroy D, Chandrashekaran V, Ryan CN, Chan LS, Testerman T, Burch J, Hofseth LJ, Horner R, Nagarkatti M, Nagarkatti P, Lasley SM, Chatterjee S. Altered gut microbiome in a mouse model of Gulf War Illness causes neuroinflammation and intestinal injury via leaky gut and TLR4 activation. PLoS One. 2017;12(3):e0172914. Epub 2017 Mar 22 PubMed.
- Seth RK, Maqsood R, Mondal A, Bose D, Kimono D, Holland LA, Janulewicz Lloyd P, Klimas N, Horner RD, Sullivan K, Lim ES, Chatterjee S. Gut DNA Virome Diversity and Its Association with Host Bacteria Regulate Inflammatory Phenotype and Neuronal Immunotoxicity in Experimental Gulf War Illness. Viruses. 2019 Oct 21;11(10) PubMed.
- Janulewicz PA, Seth RK, Carlson JM, Ajama J, Quinn E, Heeren T, Klimas N, Lasley SM, Horner RD, Sullivan K, Chatterjee S. The Gut-Microbiome in Gulf War Veterans: A Preliminary Report. Int J Environ Res Public Health. 2019 Oct 4;16(19) PubMed.
Further Reading
News
- Neurons Cave When Astrocytes Heap on the Complements
- Microglia Rely on Mixed Messages to Select Synapses for Destruction
- Paper Alert: Microglia Mediate Synaptic Loss in Early Alzheimer’s Disease
- Sans Complement: Amyloid Grows, Synapses and Memory Stay
- Nixing Complement Protein Protects Neurons in Tauopathy Model
- Soluble Aβ: Getting a Grip on Its Fate
- Two Proteins in Young Blood Give Synapses a SPARC
Primary Papers
- Horowitz AM, Fan X, Bieri G, Smith LK, Sanchez-Diaz CI, Schroer AB, Gontier G, Casaletto KB, Kramer JH, Williams KE, Villeda SA. Blood factors transfer beneficial effects of exercise on neurogenesis and cognition to the aged brain. Science. 2020 Jul 10;369(6500):167-173. PubMed.
- Mondal A, Bose D, Saha P, Sarkar S, Seth R, Kimono D, Albadrani M, Nagarkatti M, Nagarkatti P, Chatterjee S. Lipocalin 2 induces neuroinflammation and blood-brain barrier dysfunction through liver-brain axis in murine model of nonalcoholic steatohepatitis. J Neuroinflammation. 2020 Jul 4;17(1):201. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.