Amyloid Clearance: Check. Cognitive Benefit: Um … Maybe.
Quick Links
With aducanumab’s resurrection driving renewed interest in anti-amyloid antibody therapies, researchers at the 12th Clinical Trials on Alzheimer’s Disease conference, held December 4–7 in San Diego, happily provided updates on their own. Presentations on gantenerumab, donanemab, and BAN2401 reinforced previous reports that all three mop up plaques, and added more evidence that this clearance is sustained over time, even after a treatment interruption. There are hints—though that is all they are at this point in time—that amyloid clearance correlates with preserved memory, as well.
- Gantenerumab and donanemab drive plaque load to zero over time.
- Clearance with BAN2401 maintained even after a break of several years.
- Crenezumab has little effect on plaque or tangle buildup.
Among antibodies still in Phase 2 or 3, Roche’s crenezumab appears to be the outlier. In the terminated Phase 3 CREAD studies, it did not affect cognitive decline, and in San Diego, the company reported that it did not budge amyloid or tau buildup, either. However, crenezumab did lower soluble Aβ plus a marker of synaptic damage, suggesting target engagement. Overall, data at CTAD supported the idea that several antibodies are still in the running as Alzheimer’s treatments.
“I believe amyloid deserves the attention we give it,” Paul Aisen of the University of Southern California, Los Angeles, said in a keynote address. He noted that all evidence suggests the core pathological process of Alzheimer’s disease is closely linked to amyloid accumulation. After the recent signal that aducanumab probably slowed cognitive decline in one Phase 3 trial, researchers in the field are increasingly optimistic that targeting amyloid will be effective (Dec 2019 conference news).
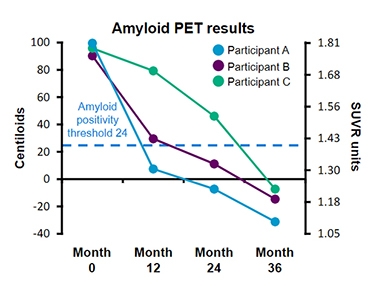
Different Trajectories, Same Result. Three representative patients in the gantenerumab long-term extension study cleared plaque at different rates, but all ended up below zero at year three. [Courtesy of Roche.]
So how well do current antibodies clean up plaque? Gregory Klein of Roche previously reported that two years of treatment with high-dose gantenerumab drove down amyloid load (Dec 2017 news; Aug 2018 news). In San Diego, Klein added three-year data from the same open-label extension study. This OLE originally enrolled 89 patients, and 30 remain in the study at this point. Ten came originally from the Scarlet RoAD trial, 12 from the Marguerite RoAD placebo group, and eight from the Marguerite RoAD treatment group.
Klein noted that the time course of amyloid removal in the OLE varied from patient to patient. He showed three representative examples of people who all started with amyloid loads of 90 to 100 centiloid (see image above). In the first, the plaque burden plummeted by about 90 centiloid in one year, falling to near-zero. Plaque clearance continued at a slower rate for the next two years, with this person now at -30 centiloid. The centiloid scale can go as low as -50. Another participant dropped from 90 centiloid to the positivity threshold of 24 in the first year, then lost plaque more gradually, ending up around -10 centiloid by the third year. The third patient started slow and sped up, clearing about 20 centiloid the first year, 30 the second, and 50 the third, to end below zero in year three. Despite this variation, overall the amyloid load fell continuously in every participant, without hitting plateaus and stalling, Klein said.
After three years, the OLE participants’ average amyloid burden is zero centiloid, representing a drop of 80 or 90 centiloid from baseline. Eighty percent of them are below the amyloid-positivity threshold, compared with 50 percent at the two-year mark. In general, people who start with a higher amyloid burden tend to lose it faster, Klein noted. Scarlet RoAD participants started the OLE with an average centiloid of 53, and ended up losing 57 over three years, while the Marguerite RoAD placebo group started at 91 and lost 90 centiloid on average.
Klein did not discuss cognitive outcomes for these people in San Diego. However, Paul Delmar of Roche had reported two-year cognitive data at AAIC last July, and the data were published on December 12 (Klein et al., 2019). At this timepoint, a regression analysis found that greater amyloid clearance correlated with slower cognitive decline. To look at the data another way, the researchers split the 39 participants remaining in the two-year OLE cohort at that time into those in whom amyloid reduction was either more, or less, than the median of 55 centiloids less than at baseline. Those in the “more-clearance” group had declined 0.8 points less on the CDR-SB, 2.2 points less on the ADAS-Cog11, and 0.9 points less on the MMSE than those in the “less-clearance” group.
These directional trends on cognition were not statistically significant in these small groups, and the authors cautioned against overinterpreting them in a diverse group that included people with prodromal and moderate AD to start with. At OLE baseline, participants had an average MMSE score of 21. When averaged across this OLE group, the cognitive benefit in the high-clearance group amounted to a 20 percent slowing of decline on the CDR-SB, 47 percent on the ADAS-Cog11, and 17 percent on the MMSE, similar in size to the signal reported in the Phase 3 aducanumab trial.
How about Eli Lilly’s donanemab? Previously known as N3pG, this Phase 2 antibody banishes plaque from the brain faster than gantenerumab does. It targets a pyroglutamate form of Aβ found only in amyloid plaques, and reportedly carries a lower risk of ARIA. The Phase 1b trial enrolled 61 participants with prodromal to moderate AD whose average baseline amyloid load was 105 centiloid. Previously, Lilly reported rapid plaque clearance with six months of dosing (Aug 2018 conference news).
In San Diego, Eli Lilly’s Stephen Lowe added 16-month data from this group. At monthly doses of 10 or 20 mg/kg, their amyloid load dropped an average of 90 to 100 centiloid. Curiously, the 10 mg/kg biweekly dose group had a smaller average reduction than either monthly group, amounting to about 50 centiloid, though by this time point, the groups were small. Only nine people remained in the 10 mg/kg biweekly dose group, six in the 10 mg/kg monthly, and five in the 20 mg/kg. In all five in the latter group, their amyloid load was below the threshold of positivity.
As in the six-month data, 26 percent of patients developed ARIA-E. Two of the 12 cases were symptomatic, with participants complaining of headache, confusion, or sleepiness that resolved after dosing was stopped. ApoE genotype did not affect ARIA or the rate of amyloid removal. Lowe noted that this trial has just completed dosing, with the last patient visit on November 5, and will undergo data lock soon. Lilly is currently testing donanemab in an 18-month Phase 2 trial, which enrolls 266 people with prodromal to mild AD.
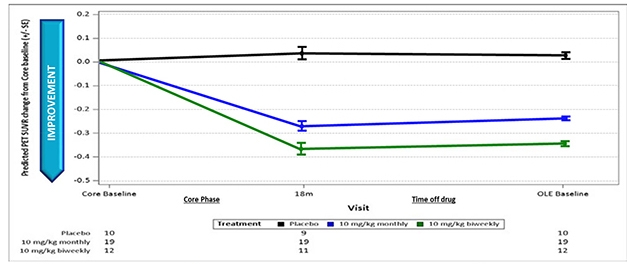
And Stay Gone! Once removed with high-dose BAN2401 (blue and green lines), amyloid plaque stays low over a gap period as long as four years (black, placebo control). [Courtesy of Eisai.]
And then there is BAN2401. The CTAD presentation on this anti-protofibril antibody discussed what happens when a person takes a break from dosing. In Phase 2, BAN2401 cleared plaque and slowed cognitive decline by a bit after 18 months of treatment (Jul 2018 news; Jul 2018 conference news; Nov 2018 conference news). This led the Eisai/Biogen partners to invite participants back to begin an open-label extension (May 2019 conference news), and in San Diego, Eisai’s Chad Swanson presented baseline data from it.
This OLE enrolled 35 people who had previously been on the 10 mg/kg biweekly dose, 52 people from the 10 mg/kg monthly group, 26 from the 5 mg/kg biweekly, six from the 5 mg/kg monthly, and 37 from the placebo groups. At OLE baseline, they had been off drug for an average of two years but with wide variability, spanning from nine months to 4½ years. Despite this range, their amyloid burden remained almost flat during this gap period. At the end of treatment, participants had a mean SUVR of 1.05, and at OLE baseline, 1.08. About 80 percent of participants were amyloid-negative by visual read at the end of Phase 2, and this remained true for those who enrolled in the OLE. Spaghetti plots of the two 10 mg/kg groups showed similar flat trajectories for nearly all participants, although in a handful of people in the monthly, but not biweekly, dose group, some amyloid returned.
By contrast, their cognition started sliding again during the gap period. There was a dose-dependence to this. For those who had received low doses, their cognitive decline caught up to that of the placebo group by the end of the gap period, with indistinguishable scores. Participants on either of the 10 mg/kg doses, on the other hand, maintained the small numerical improvement over placebo they had gained in the blinded trial. Once they went off drug, however, their cognitive decline resumed at the same rate as in the placebo group. This maintained benefit implies a disease-modifying effect, Swanson noted. Because cognitive decline resumed even while plaque load stayed low, it may be that Aβ protofibrils are doing the damage, Swanson speculated. To prevent this, people might need to remain on treatment even after their amyloid load falls below the plaque threshold, he added.
Eisai updated the CTAD audience on their design of a Phase 3 trial. Called CLARITY, it began enrolling in March 2019 and aims to pull in 1,566 people with MCI or mild AD, the same population targeted in Phase 2. They will be randomized evenly to receive placebo or 10 mg/kg biweekly for 18 months. As in Phase 2, the primary outcome measure will be the CDR-SB, with ADCOMS, ADAS-Cog14, and amyloid PET as secondary measures.
Odd One Out?
Crenezumab stood apart at CTAD, because its data showed little effect on biomarkers of pathology. This antibody recognizes soluble Aβ, with 10-fold selectivity for oligomers over monomers, and was previously reported to lower Aβ oligomers in CSF (Jul 2018 conference news). The two Phase 3 CREAD trials were halted in January, and subsequent analysis of CREAD1 data found no effect on cognitive decline (Jan 2019 news; May 2019 conference news).
In San Diego, Tobias Bittner of Roche presented biomarker findings for CREAD1, which enrolled 813 people with an average MMSE of 24. Of these, 351 took part in the amyloid PET substudy. Over two years, amyloid load increased less in people on drug than those on placebo, but the difference was not statistically significant. CREAD1’s tau PET substudy comprised 22 participants on drug and 24 on placebo. It showed a slight worsening of 0.05 SUVR in those on drug after a year, but this might be noise from the small sample size and short follow-up, Bittner suggested.
The CSF substudy comprised 139 people on drug and 140 on placebo. It showed a slight drop of total tau and p-tau181 with treatment, indicating improvement, although again this fell short of statistical significance. Numerically, total tau dropped by 10 pg/ml, p-tau181 by about two. Volumetric MRI showed no difference at all between groups.
Did anything change substantially with crenezumab? CSF Aβ40 and Aβ42 rose significantly and dose-dependently, suggesting that crenezumab hit its target in the central nervous system. Aβ42 jumped about 300 pg/ml from baseline values, and Aβ40 about 7,000. CSF Aβ falls during the progression of AD pathogenesis. CSF neurogranin, a marker of synaptic dysfunction, dropped significantly, falling by about 100 pg/ml. Four other biomarkers of neuronal injury or inflammation—NfL, α-synuclein, YKL-40, GFAP—nudged in the desired direction. Curiously, sTREM2, which has been associated with protection against decline, dropped (Aug 2019 news). Crenezumab is being tested in the API Colombian trial in a preventative paradigm (Aug 2019 conference news).
Overall, the antibody data fed the enthusiasm sparked by the aducanumab findings also shown in San Diego. Cynthia Lemere of Brigham and Women’s Hospital in Boston noted that the totality of evidence now suggests anti-amyloid antibodies will work as a preventative treatment to delay AD progression. “Now it’s a matter of refining the therapies and trying combinations,” she told Alzforum.—Madolyn Bowman Rogers
References
Therapeutics Citations
News Citations
- Exposure, Exposure, Exposure? At CTAD, Aducanumab Scientists Make a Case
- High-Dose Gantenerumab Lowers Plaque Load
- Four Immunotherapies Now Banish Amyloid From the Brain
- Topline Results: 18 Months of BAN2401 Might Work
- BAN2401 Removes Brain Amyloid, Possibly Slows Cognitive Decline
- Second Look at BAN2401 Data Still Positive, Despite Snafu
- Keep Your Enthusiasm? Scientists Process Brutal Trial Data
- On Target: Crenezumab Reduces Aβ Oligomers in CSF
- Roche Pulls Plug on Two Phase 3 Trials of Crenezumab
- In Alzheimer’s, More TREM2 Is Good for You
- Crenezumab Update: Baseline Data from Colombian Prevention Trial
Paper Citations
- Klein G, Delmar P, Voyle N, Rehal S, Hofmann C, Abi-Saab D, Andjelkovic M, Ristic S, Wang G, Bateman R, Kerchner GA, Baudler M, Fontoura P, Doody R. Gantenerumab reduces amyloid-β plaques in patients with prodromal to moderate Alzheimer's disease: a PET substudy interim analysis. Alzheimers Res Ther. 2019 Dec 12;11(1):101. PubMed.
Other Citations
External Citations
Further Reading
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.