Amyloid—It’s Not Whether, but for How Long You’ve Had It
Quick Links
Critics of the amyloid hypothesis point out that some people with brain amyloid are cognitively fine, thank you very much. Indeed, variability in plaque burden among cognitively normal older people, combined with typically short follow-up periods of a few years, have limited researchers’ ability to prove a causal relationship. Now, a new analysis suggests that it’s not so much a person’s amount of brain amyloid that determines whether they decline, but the length of time they have had it. In a manuscript posted to bioRχiv on September 23, researchers led by Sterling Johnson at the University of Wisconsin School of Medicine and Public Health, Madison, report that amyloid duration, aka “chronicity,” explains much of the variability in PET data, and correlates with abnormal cognition and the speed of cognitive decline.
- People get amyloid at different ages, muddling cause-effect studies.
- Researchers have devised a new way to estimate for how long a person has had amyloid in his or her brain.
- Duration best correlates with cognitive decline and tau pathology.
How long a person had had brain amyloid also predicted accumulation of tau in his or her entorhinal cortex. The authors claim that their algorithm can calculate chronicity from a person’s age and a single amyloid PET scan. This metric may help researchers better understand the trajectory of disease progression and could be used as a stratification criteria in clinical trials, Johnson told Alzforum. “Chronicity allows us to better put things in temporal order, figure out what are meaningful drivers of cognitive decline, and ask what factors modify these patterns,” he said.
Other researchers were enthusiastic when Johnson presented the findings at an AAIC imaging preconference last July. “This work, in effect, cleans up the noise in amyloid accumulation trajectories along a new and more functional x-axis,” noted Samuel Lockhart, Wake Forest School of Medicine, Winston-Salem, North Carolina (see comment below). “It does so by producing a chronicity variable that does a statistically superior job of predicting biomarker, cognitive, and clinical outcomes.”
“Our motivation was to see if we could estimate how long someone had been amyloid-positive based on longitudinal PET,” said joint first author Tobey Betthauser. The researchers surmised that the longer someone was exposed to the pathology, the worse off they might be, and that would illustrate how the disease progresses.
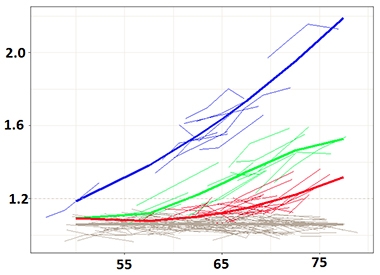
Amyloid Trajectories. Group-based trajectory modeling suggests amyloid accumulators fall into one of four groups. [Courtesy of Sterling Johnson, University of Wisconsin.]
To determine duration, Betthauser and co-first author Rebecca Koscik used a statistical algorithm called group-based trajectory modeling. GBTM was developed to understand how biological phenomena evolve over time (for review see Nagin and Odgers, 2010). Koscik used GBTM to determine trajectories of amyloid accumulation in 171 participants in the Wisconsin Registry for Alzheimer’s Prevention study. The WRAP cohort comprises middle-aged and older adults who have a family history of AD. Each of the 171 had had two to four amyloid PET scans with an average of nearly six years between first and last. Of these volunteers, 37 tested positive for brain amyloid on at least one scan.
The modeling fit volunteers into one of four categories. One comprised people who were not accumulating amyloid, while the other three groups had accumulated different amounts (see image above). Importantly, the volunteers began accumulating amyloid at different ages, but once they became positive, they all accumulated amyloid at about the same rate.
Next, the researchers were able to assign 86 additional WRAP volunteers into one of these four groups using only their ages and their amyloids load at last scan. They then used these trajectories to determine the age at which all 257 members of the cohort had become amyloid-positive. And then, by simple subtraction from age at last scan, they determined how long each person had likely been positive. A clear correlation emerged between amyloid duration and load, explaining much of the heterogeneity in amyloid burden with age (see image below).

New Context for DVR. Different people start accumulating amyloid at different ages, obscuring simple amyloid load/age correlations (left). A correlation emerges between amyloid load and chronicity (right). Colors represent the four trajectory groups. [Courtesy of Sterling Johnson.]
Is the chronicity concept useful? The researchers asked if it related to cognition and tau pathology. Chronicity better predicted decline on the PACC-3, a preclinical Alzheimer’s disease cognitive composite, than did absolute amyloid load. It also predicted levels of entorhinal tangles as determined by MK-6240 PET better than did age or simple amyloid positivity.
The authors determined that cognitively normal people who developed mild cognitive impairment or dementia had had amyloid for an average of 15 years longer than those who were stable. Chronicity could improve prediction of progression even in the prodromal phase of the disease, noted Lockhart.
“[Chronicity] is an inventive idea,” wrote Rik Ossenkoppele, Amsterdam University Medical Center, to Alzforum. “The added value of the amyloid chronicity measure compared to binarized amyloid PET results is clear, but further studies are needed to evaluate whether it also outperforms a global or regional amyloid measure,” he wrote (see comment below). Ossenkoppele noted that relying on an amyloid cutoff to determine chronicity is a disadvantage, since it overlooks the information inherent in sub-threshold levels.
The authors acknowledged that the WRAP cohort is enriched for people with an Alzheimer’s family history who have higher risk for AD than the population as a whole. “It is unlikely that our exact equations and parameters will generalize to other radiotracers and study samples,” they wrote. Still, if the concept holds up in attempts to replicate it, then chronicity may help researchers better understand risk factors for AD, and target prevention and treatment measures.—Tom Fagan
References
Paper Citations
- Nagin DS, Odgers CL. Group-based trajectory modeling in clinical research. Annu Rev Clin Psychol. 2010;6:109-38. PubMed.
External Citations
Further Reading
No Available Further Reading
Primary Papers
- Koscik RL, Betthauser TJ, Jonaitis EM, Allison SL, Clark LR, Hermann BP, Cody KA, Engle JW, Barnhart TE, Stone CK, Chin NA, Carlsson CM, Asthana S, Christian BT, Johnson SC. Amyloid duration is associated with preclinical cognitive decline and tau PET. Alzheimers Dement (Amst). 2020;12(1):e12007. Epub 2020 Feb 13 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Wake Forest School of Medicine
This manuscript by Koscik et al. on amyloid duration—or “chronicity”—presents an innovative and valuable approach to a well-trodden research question: How can we best model brain amyloid accumulation to analyze its associations with tau pathology, cognitive function, and clinical decline? As the paper notes, multiple previous studies have employed longitudinal amyloid PET data to examine progression along the AD continuum. However, this work, by using group-based trajectory modeling to “[recast] the magnitude of amyloid elevation along the time dimension,” in effect cleans up the noise in amyloid accumulation trajectories along a new and more functional x-axis. It does so by producing a chronicity variable that does a statistically superior job (relative to chronological age, continuous PiB DVR, or PiB positivity) of predicting biomarker, cognitive, and clinical outcomes.
In clearly demonstrating trajectory group membership in initially asymptomatic individuals and predicting the time to conversion to amyloid positivity in currently amyloid-negative individuals, this research helps us better understand the earliest stages of pathological amyloid accumulation. In addition, by using this chronicity measure—in particular, to quantify how long an individual has been amyloid-positive—to provide improved predictions of not only entorhinal tau pathology using MK-6240 PET but also progression to MCI and AD, this work enhances our ability to quantify disease progression and the impacts of brain amyloid deposition even in the prodromal phase of AD. Further, the aforementioned improved ability to predict clinical progression may be beneficial for disease prognosis in clinically asymptomatic individuals as well. Of course, notable limitations are the family-history enriched sample with a relatively low level of underrepresented minorities, so the results may not necessarily generalize to other, more diverse populations with different risk levels. However, this study is an important proof-of-concept work that may increase our knowledge of the early transitions from normal cognition to cognitive impairment and dementia.
VU University Medical Center
This is an interesting approach to obtain additional information from amyloid PET scans beyond dichotomization (A+/-) or a composite measure of amyloid PET signal. This fits a series of studies taking alternative approaches to increase the predictive value of amyloid PET measures for cognition or other AD biomarkers, e.g. “time-to-amyloid-positivity” (Insel et al., 2017), subthreshold amyloid PET uptake (Landau et al., 2018) or amyloid PET signal in CSF+/PET- groups (Palmqvist et al., 2017). The measure called “amyloid chronicity” reflects the modeled number of years for which an individual has been exposed to significant amyloid burden (A+) or the time it will take to become A+ in the future. This is an inventive idea. Strengths of the approach include the non-linear modelling to account for the presumed sigmoidal shape of amyloid accrual over time and the fact that amyloid chronicity could be reasonably well established based on a single amyloid PET scan. The added value of the amyloid chronicity measure compared to binarized amyloid PET results is clear, but further studies are needed to evaluate whether it also outperforms a global or regional amyloid measure (on some outcomes it showed better model fit, but not on others). An inherent disadvantage of the approach is that it still needs a threshold for amyloid-positivity and that the potential value of subthreshold levels of amyloid pathology is potentially not fully utilized. I look forward to studies applying this method to F18-labeled amyloid PET tracers and/or in other study populations.
References:
Insel PS, Ossenkoppele R, Gessert D, Jagust W, Landau S, Hansson O, Weiner MW, Mattsson N, Alzheimer's Disease Neuroimaging Initiative. Time to Amyloid Positivity and Preclinical Changes in Brain Metabolism, Atrophy, and Cognition: Evidence for Emerging Amyloid Pathology in Alzheimer's Disease. Front Neurosci. 2017;11:281. Epub 2017 May 17 PubMed.
Landau SM, Horng A, Jagust WJ, Alzheimer's Disease Neuroimaging Initiative. Memory decline accompanies subthreshold amyloid accumulation. Neurology. 2018 Apr 24;90(17):e1452-e1460. Epub 2018 Mar 23 PubMed.
Palmqvist S, Schöll M, Strandberg O, Mattsson N, Stomrud E, Zetterberg H, Blennow K, Landau S, Jagust W, Hansson O. Earliest accumulation of β-amyloid occurs within the default-mode network and concurrently affects brain connectivity. Nat Commun. 2017 Oct 31;8(1):1214. PubMed.
Make a Comment
To make a comment you must login or register.