Antisense RNA from C9ORF72 Repeats Is Likely Culprit in Patient Neurons
Quick Links
The hexanucleotide repeats in the C9ORF72 gene that cause amyotrophic lateral sclerosis and frontotemporal dementia undergo transcription and translation in both the sense and antisense directions. The backward RNAs and peptides outnumber the sense varieties in the motor neurons of people who died of ALS, according to a paper in the May 6 Acta Neuropathologica online. More telling, the antisense RNA was more often accompanied by mislocalized TDP-43, a hallmark of ALS and some FTDs. “This suggests the antisense [RNA] foci hold prime sway in the pathogenesis of these motor neurons,” said study first author Johnathan Cooper-Knock of the University of Sheffield, U.K.
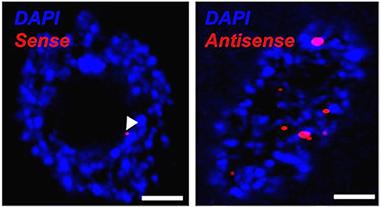
Hitting reverse.
Antisense C9ORF72 hexanucleotide RNA foci predominate in ALS motor neurons. [Image courtesy of Johnathan Cooper-Knock/Acta Neuropathologica.]
GGGGCC repeats in the C9ORF72 gene, sometimes numbering in the thousands, are the most common genetic cause of ALS and FTD (see Sep 2011 news). Both sense and antisense transcripts containing these stuttering repeats assemble into abnormal aggregates. While it’s yet unclear if these RNA foci are toxic, they serve as a marker for cells with abundant repeat RNA. Moreover, ribosomes translate those RNAs into five distinctive dipeptides, which are also potential, but unconfirmed, toxic species (see Nov 2013 news).
Previously, Cooper-Knock and senior author Pam Shaw had found sense-direction RNA foci in tissue from C9ORF72 expansion carriers that had been stored in the University of Sheffield’s brain bank. The samples contained another common ALS and FTD pathology, cytoplasmic aggregates of the normally nuclear TDP-43, but these did not correlate with the presence or absence of repeat sense RNA foci (Cooper-Knock et al., 2014). Now, Cooper-Knock and colleagues have turned their attention to antisense foci, and they observed a key difference from the sense version. In brain samples from eight people who died of C9ORF72-based ALS or FTD, the majority of motor neurons with antisense C9ORF72 RNA foci also were marked by mislocalized TDP-43.
Why would antisense but not sense RNA affect TDP-43? Cooper-Knock first assumed that the RNAs might bind different proteins, which could influence TDP-43 dynamics, but in pull-down and crosslinking experiments both RNAs turned out to interact with similar partners.
Next, Cooper-Knock theorized that perhaps the amount of RNA was critical for TDP-43 to accumulate in the cytoplasm. He counted RNA foci in five types of neurons in the brain samples: cerebellar Purkinje and granule, spinal cord motor, hippocampal dentate gyrus, and CA4 neurons. Examining hundreds of neurons, Cooper-Knock observed a consistent relationship between sense and antisense foci in each cell type. Purkinje and motor neurons always possessed more antisense than sense foci, while granule neurons consistently contained more sense foci. In the hippocampus, the two types were present in equal amounts. Because motor neurons seem inclined to transcribe antisense repeats, this may be why they are prone to mislocalize TDP-43, Cooper-Knock concluded.
He found a similar pattern when he examined dipeptide inclusions in granule and motor neurons. The sense-direction peptides only occurred in granule neurons, while the antisense versions only appeared in motor neurons. Cooper-Knock did not correlate dipeptides with TDP-43 aggregation. Jiou Wang of Johns Hopkins University in Baltimore, who was not involved in the study, thought the findings quite plausible. "The researchers examined many cells and the statistics seemed sound,” he told Alzforum.
Why do motor neurons make more antisense foci than sense foci? “It is puzzling that the expression of the antisense transcript might exceed expression of the sense transcript, given that the sense transcript utilizes canonical transcription start sites,” commented Paul Taylor of St. Jude Children’s Research Hospital in Memphis, Tennessee, who was not involved in the study. Cooper-Knock believes that motor neurons probably express some transcription factors that favor the antisense direction. Alternatively, he added, different cells may degrade the two RNAs differently, with motor neurons allowing antisense RNAs to remain.
Based on the correlations he observed, Cooper-Knock suggested that motor neurons overproduce antisense foci, which leads to excess antisense peptides, mislocalization of TDP-43, and neurodegeneration. He cannot prove this idea with only the snapshot of pathology from autopsy tissue. To understand the time course between these four steps, Cooper-Knock hopes to experiment with mouse models of C9ORF72 pathology—the first of which was just published (Chew et al., 2015).
In the meantime, he had advice for researchers developing anti-C9ORF72 therapeutics. Scientists may need to target the antisense repeats, to achieve an effective treatment, he said.—Amber Dance
References
News Citations
- Corrupt Code: DNA Repeats Are Common Cause for ALS and FTD
- Sense, Antisense: C9ORF72 Makes Both Forms of RNA, Peptides
Paper Citations
- Cooper-Knock J, Walsh MJ, Higginbottom A, Robin Highley J, Dickman MJ, Edbauer D, Ince PG, Wharton SB, Wilson SA, Kirby J, Hautbergue GM, Shaw PJ. Sequestration of multiple RNA recognition motif-containing proteins by C9orf72 repeat expansions. Brain. 2014 Jul;137(Pt 7):2040-51. Epub 2014 May 27 PubMed.
Further Reading
Papers
- Lee YB, Chen HJ, Peres JN, Gomez-Deza J, Attig J, Stalekar M, Troakes C, Nishimura AL, Scotter EL, Vance C, Adachi Y, Sardone V, Miller JW, Smith BN, Gallo JM, Ule J, Hirth F, Rogelj B, Houart C, Shaw CE. Hexanucleotide repeats in ALS/FTD form length-dependent RNA foci, sequester RNA binding proteins, and are neurotoxic. Cell Rep. 2013 Dec 12;5(5):1178-86. Epub 2013 Nov 27 PubMed.
- Mackenzie IR, Arzberger T, Kremmer E, Troost D, Lorenzl S, Mori K, Weng SM, Haass C, Kretzschmar HA, Edbauer D, Neumann M. Dipeptide repeat protein pathology in C9ORF72 mutation cases: clinico-pathological correlations. Acta Neuropathol. 2013 Dec;126(6):859-79. Epub 2013 Oct 6 PubMed.
- Sareen D, O'Rourke JG, Meera P, Muhammad AK, Grant S, Simpkinson M, Bell S, Carmona S, Ornelas L, Sahabian A, Gendron T, Petrucelli L, Baughn M, Ravits J, Harms MB, Rigo F, Bennett CF, Otis TS, Svendsen CN, Baloh RH. Targeting RNA Foci in iPSC-Derived Motor Neurons from ALS Patients with a C9ORF72 Repeat Expansion. Sci Transl Med. 2013 Oct 23;5(208):208ra149. PubMed.
Primary Papers
- Cooper-Knock J, Higginbottom A, Stopford MJ, Highley JR, Ince PG, Wharton SB, Pickering-Brown S, Kirby J, Hautbergue GM, Shaw PJ. Antisense RNA foci in the motor neurons of C9ORF72-ALS patients are associated with TDP-43 proteinopathy. Acta Neuropathol. 2015 Jul;130(1):63-75. Epub 2015 May 6 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Cooper-Knock and colleagues report the presence of increased antisense foci in motor neurons relative to sense foci. There are a few possible reasons why this may occur:
1) It is possible that there is increased transcription of antisense RNA relative to sense RNA. It would be interesting to see a comprehensive analysis done on both control and C9 patients in different tissues to measure the relative rates of transcription of both the sense and antisense RNA levels. There might be cell-type specific transcriptional proteins that regulate the transcription of sense vs. antisense RNA.
In my view, understanding how high levels of antisense RNA are generated in C9ORF72 patient cells is an open question and needs to be explored further.
2) A second possibility would be that the sense and antisense strands maintain different conformations or stabilities. It has been reported previously that the sense strand forms G-quadruplexes whereas the antisense strand does not (Reddy et al., 2013). The stability/conformation of these RNAs might be regulated in a cell-specific manner. It would be interesting to know what the half-life of each RNA species is and whether this varies by cell type.
References:
Reddy K, Zamiri B, Stanley SY, Macgregor RB, Pearson CE. The disease-associated r(GGGGCC)n repeat from the C9orf72 gene forms tract length-dependent uni- and multimolecular RNA G-quadruplex structures. J Biol Chem. 2013 Apr 5;288(14):9860-6. PubMed.
Make a Comment
To make a comment you must login or register.