Does Aβ Come In Strains? Glimpse Into Human Brain Suggests Yes
Quick Links
In animal studies, aggregates of amyloid β seem to act like prions, forming specific strains that spread through the brain from an initial seed. Now, in the September 12 Cell, researchers led by Robert Tycko at the National Institutes of Health, Bethesda, Maryland, suggest the same thing happens in people. The researchers spiked solutions of monomeric Aβ40 with extracts from the brains of two Alzheimer’s disease patients to seed growth of fibrils. They found that fibrils grown from amyloid taken from multiple brain regions of the same patient had identical structures. However, fibrils from the two patients varied markedly from each other, and also from fibrils grown directly from monomers in vitro. “This is the first concrete evidence of Aβ strains in patients,” Tycko told Alzforum. Nonetheless, it remains to be proven that the fibrils expanded in vitro are identical to the starting material in brain. Tantalizingly, the two patients had quite different clinical presentations, but it is unclear if those symptoms correlate with fibril structure.
Other researchers lauded the data, which provide the first detailed look at the potential architecture of amyloid fibrils from patient brains. The findings may not only help researchers better understand how amyloid forms, but may also help them design therapeutics. “These structural insights offer a treasure trove for the structure-based design of antagonists of Aβ aggregation,” wrote Adriano Aguzzi at University Hospital Zurich, Switzerland, and Aaron Gitler at Stanford University, California, in an accompanying editorial. Other researchers noted that data from many more patients will be needed to understand how these findings might relate to disease.
The idea that Aβ, and other amyloidogenic proteins, can form distinct strains has been debated for some time. Most of the evidence has come from animal work. In fact, in the September 3 EMBO Reports, researchers led by Mathias Jucker at the University of Tübingen, Germany, reported that brain extracts from AD transgenic mice make distinct Aβ strains. First author Götz Heilbronner injected brain material from aged, plaque-laden APP/PS1 or APP23 transgenic AD mice into young APP23 mice. The young mice developed plaques that had the structural characteristics of the source material. Some of this work was previously published (see ARF related news story). “The evidence for transmissible amyloid conformers (i.e. strains) is now overwhelming,” Jucker wrote to Alzforum.
In previous work, Tycko and colleagues found that Aβ generates a wide variety of structures in vitro, depending on the exact aggregation conditions (see Petkova et al., 2005). These strains vary in toxicity and perpetuate themselves through self-seeding. The authors wondered if the same types of structure grow in human brains. To begin to answer this, they developed methods for extracting amyloid-enriched samples from postmortem brain tissue (see Paravastu et al., 2009). In the current paper, first author Jun-Xia Lu refined the techniques, using gentler methods so as not to disrupt native fibrils. Brain samples seeded synthetic, isotopically-labeled Aβ40. This approach produced a high enough quantity of fibrils to analyze with nuclear magnetic resonance (NMR) spectroscopy. Because NMR is highly sensitive to small variations in structure, the resulting spectra serve as a “fingerprint” of a given molecule.
Samples from either the occipital or temporal/parietal lobe of “patient 1” produced Aβ40 fibrils with an identical fingerprint. Likewise, Aβ40 monomers seeded with occipital, frontal, or temporal lobe material of “patient 2” grew fibrils that matched each other, but differed from those from patient 1. Importantly, tissue from non-AD control brains seeded no fibrils. The authors extensively characterized the fine structure of aggregates from patient 1 using NMR and electron microscopy. The fibrils exhibited a three-fold symmetry around the long axis, created by three separate Aβ peptides interacting (see image below). The strands also boasted a more complicated structure than fibrils formed in vitro, with unique kinks and bends. The structural differences between fibrils bury or expose different amino acid side chains, which could have large consequences for how fibrils interact with other molecules such as diagnostic or therapeutic agents, the authors note.
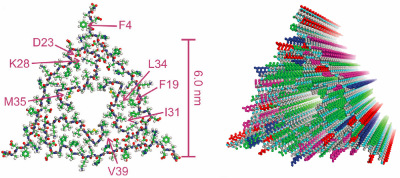
Fibrils from an AD patient display a threefold symmetry around the long axis. Image courtesy Robert Tycko and Cell Press.
Intriguingly, the two patients had very different symptoms. While postmortem autopsy revealed typical Alzheimer’s pathology of neuritic plaques and neurofibrillary tangles in both, patient 1 had been clinically diagnosed with Lewy Body Disorder and patient 2 with AD. Patient 1 had difficulty finding words, and her brain had only mild atrophy in frontal and parietal lobes. By contrast, patient 2 had memory impairments typical of AD and gross brain atrophy and gliosis. In ongoing work, Tycko and colleagues are analyzing fibrils grown from many more patients to see if fibril structure consistently correlates with pathology and clinical symptoms. If that were true, amyloid strains could become very important for categorizing AD patients and perhaps determining prognosis and treatment, the authors speculate. Tycko also wants to compare fibrils made from asymptomatic older adults with brain amyloid to see if their fibrils differ structurally from those generated by AD patients. In addition, he is analyzing Aβ42 fibrils. Notably, the authors did not examine smaller Aβ aggregates such as oligomers, which many researchers now believe to be the most toxic forms. It is unclear if the structure of oligomers correlates with that of fibrils; some studies suggest they are quite different (see ARF related news story).
Because Aβ forms many different fibril structures in vitro based on slight variations in conditions, it seems unlikely that fibrils in different brain regions of the same patient would spontaneously develop in the same way, the authors suggest. Therefore, the finding of a uniform structure throughout brain suggests that amyloid fibrils arise from an initial nucleation event and then propagate. Several previous mouse studies have shown that amyloid can spread through the brain from a single seed (see ARF related news story; ARF related news story).
Researchers agree that more evidence is needed to prove this occurs in human brain, however. Commentators wondered whether the amplified fibrils faithfully reflect those present in brain. Marc Diamond at Washington University in St. Louis, Missouri, who has propagated tau strains from human brain in cell cultures (see ARF related news story), noted that even small changes in the environment, such as a shift in temperature, can result in different structures forming. David Teplow at the University of California, Los Angeles, speculated that multiple strains could be present in AD brain, but perhaps only one can be amplified by the seeding protocol.
Nonetheless, researchers agreed that the structural studies were meticulously done and that this first glimpse at the precise architecture of human amyloid will be of great value. In their editorial, Aguzzi and Gitler note that this data may help researchers to develop amyloid binding ligands for positron emission tomography that are specific for subsets of patients. In the handful of patients who carry the Arctic APP mutation, current ligands cannot detect amyloid deposits (see ARF related news story). There are also a substantial number of patients, dubbed SNAP (for suspected non-amyloid pathology), who have typical clinical signs of AD without any apparent brain amyloid (see ARF related news story). One possibility is that SNAP patients have a form not detected by current PET and postmortem pathology labels. The structural information could also guide the development of antibodies against amyloid, Aguzzi and Gitler suggest. If different Aβ morphologies are selectively targeted by subsets of anti-Aβ antibodies, then the limited success of current AD immunotherapy may be due, at least in part, to inadequate patient stratification, they speculate.—Madolyn Bowman Rogers
References
News Citations
- Double Paper Alert—A Function for BACE, a Basis for Amyloid
- X-raying Aβ Oligomers, Unraveling Mysteries of Amyloid Aggregation
- Seeds of Destruction—Prion-like Transmission of Sporadic AD?
- Aβ Sufficient for Seeding—But Is It a Prion?
- Miami: When Does Amyloid Deposition Start in Familial Alzheimer’s?
- Suspected Non-Amyloid Pathology (SNAP)—Not an Open or Shut Case
Paper Citations
- Petkova AT, Leapman RD, Guo Z, Yau WM, Mattson MP, Tycko R. Self-propagating, molecular-level polymorphism in Alzheimer's beta-amyloid fibrils. Science. 2005 Jan 14;307(5707):262-5. PubMed.
- Paravastu AK, Qahwash I, Leapman RD, Meredith SC, Tycko R. Seeded growth of beta-amyloid fibrils from Alzheimer's brain-derived fibrils produces a distinct fibril structure. Proc Natl Acad Sci U S A. 2009 May 5;106(18):7443-8. PubMed.
Other Citations
Further Reading
No Available Further Reading
Primary Papers
- Lu JX, Qiang W, Yau WM, Schwieters CD, Meredith SC, Tycko R. Molecular Structure of β-Amyloid Fibrils in Alzheimer's Disease Brain Tissue. Cell. 2013 Sep 12;154(6):1257-68. PubMed.
- Aguzzi A, Gitler AD. A Template for New Drugs against Alzheimer's Disease. Cell. 2013 Sep 12;154(6):1182-4. PubMed.
- Heilbronner G, Eisele YS, Langer F, Kaeser SA, Novotny R, Nagarathinam A, Aslund A, Hammarström P, Nilsson KP, Jucker M. Seeded strain-like transmission of β-amyloid morphotypes in APP transgenic mice. EMBO Rep. 2013 Oct 30;14(11):1017-22. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Edinburgh
This interesting paper implies that while amyloid fibrils appear to have a uniform structure in the plaque of a given patient with Alzheimer’s disease (AD), that structure may differ from patient to patient. This conclusion is based on data from only two people, but assuming it holds up in more cases, it could be important for our understanding of people's vulnerability (or, conversely, resistance) to developing sporadic AD. Together, this data and the recent paper by Cohen et al. showing that fibrils are important for the assembly of toxic oligomers (see ARF related news story) could mean that each individual's fibril type may have a greater or lesser capability to seed toxic oligomers, which may account for some people being more resistant to AD despite a high plaque load.
References:
Cohen SI, Linse S, Luheshi LM, Hellstrand E, White DA, Rajah L, Otzen DE, Vendruscolo M, Dobson CM, Knowles TP. Proliferation of amyloid-β42 aggregates occurs through a secondary nucleation mechanism. Proc Natl Acad Sci U S A. 2013 Jun 11;110(24):9758-63. PubMed.
View all comments by Tara Spires-JonesTara Spires-Jones’ comment above regarding the potential importance of variation in amyloid structure from patient to patient is certainly reasonable. However, another and perhaps more likely explanation for the different results obtained by Lu et al. is that the extracts from which seed materials were derived contained factors or chemically modified proteins that altered fibril structure. For example, it is unclear what fraction of methionine residues in the seed material was oxidized. Small differences, such as methionine oxidation or other covalent modifications, may lead to profoundly different structures when such seeds are used to induce fibrillization of synthetic material.
Another concern is that Lu et al. generated the proposed three-dimensional fibril structure using relatively few geometric constraints (listed only in supplementary tables). NMR-determined protein structures are often generated with several constraints per residue. In this case, however, there were barely more than half as many constraints as residues, and most residues had no constraints at all. Models generated with so few constraints must be regarded as “underdetermined,” which implies that the final computer-generated model will be strongly influenced by the starting structure to which the constraints are applied. In this context, it seems remarkable that there is no mention of the "staggered" relationship between the two halves of the peptide described in earlier publications from this lab.
I would place greater value on the findings if the seed material was shown to be predominantly unmodified Aβ40, and if greater efforts were made to show that the final structure was independent of assumptions made about the starting structure.
View all comments by Allen KennedyNIDDK, NIH
In response to Dr. Kennedy's comments:
1. Our structural model for Aβ40 fibrils from patient 1, described in this paper, was calculated with Xplor-NIH software using 197 NMR-based experimental structural restraints (4.925 restraints per residue on average), including torsion angle restraints derived from NMR chemical shifts; torsion angle and distance restraints derived from quantitative measurements of 13C-13C, 15N-13C, and 15N-15N dipole-dipole couplings; and semi-quantitative restraints on inter-residue distances derived from several types of two-dimensional solid-state NMR spectra. Structure calculations began with completely random structures (not from any specific assumed starting point). Symmetry restraints, which are dictated by the experimental data, were imposed during structure calculations. A very large number of structure calculations were performed over a period of many months, with various modifications of the Xplor-NIH scripts. None of these calculations produced structures that were significantly different from the structures we have deposited in the Protein Data Bank as PDB 2M4J (and also consistent with all experimental restraints). Full details of the experimental data and structure calculations are given in this paper.
2. I do not know the basis of Dr. Kennedy's comment that, "In this case, however, there were barely more than half as many constraints as residues, and most residues had no constraints at all." Perhaps he is focusing on one particular type of restraint, whereas in fact we used several types of restraints from several types of solid-state NMR (and electron microscopy) measurements.
3. The issue of "stagger" in amyloid structures is discussed in our earlier papers (Petkova et al., 2006; Paravastu et al., 2008). Stagger means that interactions between amino acid side chains of two different β-strands in an amyloid structure are displaced along the fibril axis, making these interactions intermolecular rather than intramolecular in nature. In the case of Aβ40 fibrils from patient 1, we were unable to test for stagger because this would have required additional fibril samples with different isotopic labeling, and we did not have sufficient tissue from patient 1 to prepare the requisite samples. For examples of staggering in Aβ40 fibrils, see PDB files 2LMN, 2LMO, 2LMP, and 2LMQ. (Note that the direction of stagger is very difficult or impossible to determine experimentally, which is why these PDB files include both positive and negative stagger.)
4. Earlier work by others has shown that Aβ40 and Aβ42 in brain tissue is chemically modified in various ways to various degrees. The bulk of these chemical modifications presumably occur gradually over time, after the fibrils are formed (although certain modifications of a fraction of the peptides may accelerate the initial nucleation of fibrils). Nonetheless, we have found that fibrils from brain tissue are capable of seeding unmodified Aβ40 and Aβ42 efficiently. At this point, we have no evidence that chemical modifications of Aβ40 in brain tissue affect the results in this paper, although this is an interesting suggestion. In our hands, oxidation of Met35 impedes fibril formation; peptides with oxidized Met35 are excluded from fibrils formed by unoxidized peptides.
References:
Petkova AT, Yau WM, Tycko R. Experimental constraints on quaternary structure in Alzheimer's beta-amyloid fibrils. Biochemistry. 2006 Jan 17;45(2):498-512. PubMed.
Paravastu AK, Leapman RD, Yau WM, Tycko R. Molecular structural basis for polymorphism in Alzheimer's beta-amyloid fibrils. Proc Natl Acad Sci U S A. 2008 Nov 25;105(47):18349-54. PubMed.
View all comments by Robert TyckoMake a Comment
To make a comment you must login or register.