Human ApoE Antibody Nips Mouse Amyloid in the Bud
Quick Links
Apolipoprotein E4 is the strongest genetic risk for late-onset Alzheimer’s disease, yet efforts to make a therapy against it have not paid off. That may be about to change. David Holtzman and colleagues at Washington University in St. Louis have generated monoclonal antibodies that selectively bind to aggregated ApoE. While ignoring the lipidated forms of ApoE that are abundant in blood and brain, these antibodies reduced amyloid plaques in mice. This clearance seems to require activation of microglia. The findings, reported in the March 30 Journal of Clinical Investigation, suggest that plaque-targeting ApoE immunotherapy might work for AD.
- New antibody binds aggregated, plaque-associated ApoE.
- In mice it stimulates microglia to suppress amyloid accumulation.
- It leaves lipidated ApoE alone.
“It’s exciting that they found an antibody selective for an apparently aggregated form of ApoE,” said Cynthia Lemere, Brigham and Women’s Hospital in Boston. “Previously, targeting ApoE seemed to be a shot in the dark, and one with potentially serious side effects. Targeting what appears to be a more pathologic form seems to be a wiser, safer strategy, and I’m excited to see they’re moving forward,” Lemere told Alzforum. Gary Landreth, Indiana University, Indianapolis, agreed. “The ability to selectively target deposited, non-lipidated forms of ApoE as a means of lowering plaque burden is therapeutically attractive, given the importance of ApoE-mediated lipid transport in the brain,” he wrote to Alzforum (see full comment below).
Apolipoprotein E plays a major role in shuttling fats around the body and cholesterol in brain, and interfering with those functions could have catastrophic consequences. Nevertheless, the E4 isoform accelerates aggregation of Aβ, impedes its clearance, and co-deposits with Aβ, making it an obvious target for a therapeutic. The Holtzman lab previously found that injecting an antibody to mouse ApoE prevented plaque formation, shrank existing plaques, and improved neuronal function and behavior in APP/PS1 mice (May 2014 news).
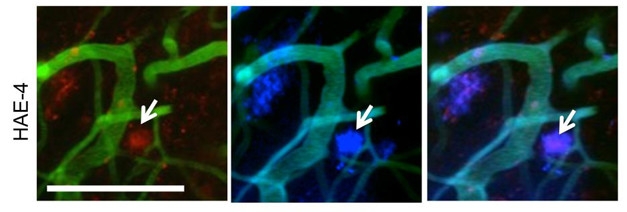
Live Action. The HAE-4 anti-ApoE antibody (red, left panel) stains amyloid plaques (blue, center) in the brain of a living mouse. Third panel shows merged image. [Courtesy of Liao et al., JCI 2018.]
To see if the same held true for human ApoE, joint first authors Fan Liao and Aimin Li generated four new monoclonal antibodies to recombinant ApoE4, then tested their ability to prevent amyloid deposition in APP/PS1 animals that had been crossed with ApoE knock-in mice. The latter express the human E4 allele in place of mouse ApoE. Liao and colleagues injected the antibodies intraperitoneally into female APPPS1/APOE4 mice once a week for seven weeks, starting when they were two months old. When the mice reached 3.5 months, the researchers measured amyloid deposition. The best of the antibodies, HAE-4, caused a 50 percent reduction in Aβ plaque load compared with an isotype-matched IgG2ab control. The HAE-4 treated mice also had reductions, albeit smaller, in insoluble brain Aβ40 and Aβ42, but no change in plasma or brain ApoE4 levels.
Continuously infusing HAE-4 into the brain reduced plaque load, as well. Given this chronic dosing, the researchers were surprised to find overall ApoE levels held steady. They soon understood why. HAE-4, which recognizes recombinant ApoE3 and E4, did not bind lipidated human ApoE in plasma from E4 or E3 knock-in mice. Instead, the antibody preferentially reacted with aggregated species of non-lipidated ApoeE4 or E3 in vitro, and with ApoE localized to amyloid plaques in the APPS1/APOE4 crosses [see image above].
How does HAE-4 reduce plaques? Intraperitoneal injection of the antibody every three days for two weeks increased the number of cells in the brain expressing CD45, a marker of activated microglia. When the authors used adenovirus to express HAE-4 in the brain with and without its microglia-targeting Fc effector domain, only the intact antibody caused reductions in plaque, fibrillar Aβ, and insoluble Aβ40/42. “We found that for antibodies to work, they need to be able to activate microglia to gobble up the plaques,” said Holtzman. In an accompanying editorial in JCI, David Borchelt, University of Florida, Gainesville, speculated that the antibody may slow or prevent plaque formation by clearing small ApoE/Aβ aggregates that seed plaques.
Mary Jo LaDu of the University of Illinois, Chicago, said the work raises many questions. Most importantly, does the antibody improve behavior? “They show reduced amyloid deposition, which may or may not be bad, and microglial activation, which may or may not be good. But in the end, we need to know if the antibody’s making these mice better,” said LaDu. And although HAE-4 binds to plaques in the mice, LaDu would like to know if it clears them. In Holtzman’s previous work, mouse ApoE antibodies both reduced existing plaque and improved behavior in APP-PS1 mice, but it remains to be seen if human antibodies do the same, he said.
Yadong Huang, Gladstone Institutes, San Francisco, was intrigued by the study. “It offers proof of concept that we can target ApoE for AD,” he said. But he, too, had questions. What epitope does the antibody recognize? And will it bind plaques in the human brain, or ApoE in plasma? Holtzman told Alzforum that HAE-4 binds plaques in human brain sections in the few cases they have examined so far.
Could ApoE antibodies offer advantages over the Aβ antibodies currently in Phase 3 trials? For one, they might have fewer side effects, such as inflammation and microhemorrhages [ARIA], although that remains to be seen, said Holtzman. It is also possible that removing non-lipidated ApoE may have benefits beyond amyloid reduction. For example, Holtzman’s group recently showed that ApoE4 contributes to tau toxicity, independent of its effects on amyloid (Sep 2017 news).
For next steps, Holtzman said they will study other mouse models, comparing HAE-4 and anti-Aβ antibodies head to head to see which can best improve behavior, neuronal and vascular function, and to look for potential side effects. Washington University has licensed the antibodies to Denali Therapeutics of South San Francisco, and Holtzman is collaborating with the company to further characterize them.—Pat McCaffrey
References
Research Models Citations
News Citations
Further Reading
No Available Further Reading
Primary Papers
- Liao F, Li A, Xiong M, Bien-Ly N, Jiang H, Zhang Y, Finn MB, Hoyle R, Keyser J, Lefton KB, Robinson GO, Serrano JR, Silverman AP, Guo JL, Getz J, Henne K, Leyns CE, Gallardo G, Ulrich JD, Sullivan PM, Lerner EP, Hudry E, Sweeney ZK, Dennis MS, Hyman BT, Watts RJ, Holtzman DM. Targeting of nonlipidated, aggregated apoE with antibodies inhibits amyloid accumulation. J Clin Invest. 2018 May 1;128(5):2144-2155. Epub 2018 Mar 30 PubMed.
- Borchelt DR. Targeting the accomplice to thwart the culprit: a new target for the prevention of amyloid deposition. J Clin Invest. 2018 May 1;128(5):1734-1736. Epub 2018 Mar 30 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
OHSU
As shown by Bales et al. in 1999, ApoE is critical for amyloid deposition; in APP(V717F) transgenic mice on a murine ApoE knockout background, the amount of Aβ40 and Aβ42 immunoreactive deposits and astrogliosis and microgliosis is markedly reduced. Consistent with these results, the Holtzman group published a paper in 2014 in which an anti-mouse ApoE antibody (HJ6.3) was used before and after plaque onset (Liao et al., 2014). When administered after plaque onset, plaque load was reduced. This was associated with a slight improvement in ability to locate a hidden platform in the water maze. The current paper is a logical follow-up study to determine the effects of antibodies against human ApoE in mice expressing both human ApoE and human APP.
In this very thorough and elegant study, the authors show that peripherally administered antibodies (HAE-4) against ApoE3 and ApoE4 preferentially bind non-lipidated, aggregated ApoE, as compared to lipidated ApoE. HAE-4 binds ApoE in amyloid plaques in unfixed brain sections and in living APPPS1-21/E4 bigenic mice, and reduces Aβ plaque load and insoluble Aβ40 and Aβ42 in the guanidine fraction of the cerebral cortex, all without affecting plasma ApoE or Aβ levels. Using adeno-associated virus to express the antibodies in the brain revealed that the Fcg receptor is required for these effects. The number of CD45-positive, but not of Iba1-positive, cells around plaques was increased following HAE-4 treatment, suggesting that the microglia present are in a more activated state.
Interestingly, weekly intraperitoneal injections of the antibodies at 50 mg/kg were more effective than continuous intracerebroventricular infusion of the antibodies at 0.3 μg/hr for six weeks. A critical question is whether this anti-ApoE antibody treatment is beneficial with regard to cognitive impairment in the mice. As we reported, plaque-independent cognitive impairments are seen in six-month-old E4/APP, but not E3/APP, bigenic mice prior to onset of plaque pathology, even though there are no differences in Aβ levels (either of Aβ1-x, as an approximate of total Aβ levels, or of Aβ42) between E3/APP and E4/APP mice (Raber et al., 2000). The requirement of activated microglia for the effect of HAE-4 on amyloid pathology is also important for addressing another related critical question: How do colony-stimulating factor 1 receptor (CSF1R) antagonists, such as PLX3397, that target microglia improve cognitive performance and rescue dendritic spine loss in 5xfAD mice without affecting amyloid pathology (Spangenberg et al., 2016)? Clearly, more research is needed to answer these questions.
References:
Bales KR, Verina T, Cummins DJ, Du Y, Dodel RC, Saura J, Fishman CE, DeLong CA, Piccardo P, Petegnief V, Ghetti B, Paul SM. Apolipoprotein E is essential for amyloid deposition in the APP(V717F) transgenic mouse model of Alzheimer's disease. Proc Natl Acad Sci U S A. 1999 Dec 21;96(26):15233-8. PubMed.
Liao F, Hori Y, Hudry E, Bauer AQ, Jiang H, Mahan TE, Lefton KB, Zhang TJ, Dearborn JT, Kim J, Culver JP, Betensky R, Wozniak DF, Hyman BT, Holtzman DM. Anti-ApoE antibody given after plaque onset decreases Aβ accumulation and improves brain function in a mouse model of Aβ amyloidosis. J Neurosci. 2014 May 21;34(21):7281-92. PubMed.
Raber J, Wong D, Yu GQ, Buttini M, Mahley RW, Pitas RE, Mucke L. Apolipoprotein E and cognitive performance. Nature. 2000 Mar 23;404(6776):352-4. PubMed.
Spangenberg EE, Lee RJ, Najafi AR, Rice RA, Elmore MR, Blurton-Jones M, West BL, Green KN. Eliminating microglia in Alzheimer's mice prevents neuronal loss without modulating amyloid-β pathology. Brain. 2016 Apr;139(Pt 4):1265-81. Epub 2016 Feb 26 PubMed.
Tel Aviv University
This study, from the Holtzman group, shows that intracerebroventricular and intraperitoneal administration of an anti-human ApoE antibody, HAE-4, which binds similarly to ApoE3 and ApoE4, reduces Aβ plaques and Aβ accumulation in brains of APPPS1/ApoE4 mice. Antibody HAE-4 binds to senile plaques as well as to aggregated and non-aggregated forms of the recombinant ApoE3 and ApoE4 proteins. However, it does not bind to the lipidated serum ApoE forms of these proteins. Furthermore, this antibody does not alter the levels of total ApoE in the brain or plasma. Additional experiments utilizing adeno-associated virus expressing normal and mutated HAE-4 antibodies revealed that the HAE-4-driven decreased amyloid accumulation depends on Fcɤ receptor function. These findings are of great importance and have general and wide implications in that they show that the antigenicity of ApoE differs between the peripheral and brain sides of the BBB. Moreover, antibodies, such as those presently employed, can be used for the development of brain anti-Aβ plaque immunotherapy and, in principle, to target any other brain-specific ApoE epitopes and pathologies.
It is of importance to note that antibody HAE-4 reacts similarly with recombinant ApoE4 and ApoE3 in vitro, and that its anti Aβ-plaque efficacy was studied in vivo utilizing APPPS1/ApoE4 mice. It would be of great interest to compare the anti-Aβ and anti-plaque efficacy of HAE-4 and similar antibodies in APPPS1 mice crossed with ApoE3, ApoE4, or heterozygous ApoE3/E4 mice. Such studies are expected to unravel the relative potency of the presently proposed approach as both anti-Aβ-plaque therapy and anti-apoE4 treatment.
A growing body of evidence suggests that the effects of ApoE4 are also mediated via Aβ-independent mechanisms such as synaptic dysfunction (Liu et al., 2013). In this context we recently showed that such effects (e.g., synaptic impairments and the accumulation of hyperphosphorylated tau and neuronal Aβ in hippocampal neurons of ApoE4-targeted replacement mice) can be counteracted by anti-ApoE immunotherapy following peripheral injection of an anti-ApoE4 antibody raised against an ApoE4-specific sequence (Luz et al., 2016). This treatment also had no effect on the overall levels of brain and peripheral ApoE, suggesting that similar to the present study, it was driven by effects of the ApoE4 antibodies on distinct brain-specific ApoE4 epitopes.
Development of anti-ApoE4 immunotherapy has been hampered by the argument that, since the concentration of ApoE in the serum is much higher than in the brain, peripherally applied anti-ApoE4 will be titered in the periphery prior to reaching the brain. The present demonstration, by the Holtzman group, that the structure and antigenicity of ApoE differs between the periphery and the brain now paves the way for development of ApoE immunotherapy targeted at Aβ pathology as well as at other ApoE4-driven pathologies.
References:
Liu CC, Liu CC, Kanekiyo T, Xu H, Bu G. Apolipoprotein E and Alzheimer disease: risk, mechanisms and therapy. Nat Rev Neurol. 2013 Feb;9(2):106-18. Epub 2013 Jan 8 PubMed.
Luz I, Liraz O, Michaelson DM. An Anti-apoE4 Specific Monoclonal Antibody Counteracts the Pathological Effects of apoE4 In Vivo. Curr Alzheimer Res. 2016 Jun 2;13(8):918-29. PubMed.
University of British Columbia
Holtzman and colleagues have made a spectacular advance toward a therapeutic approach to ApoE, which is the major genetic risk factor for Alzheimer’s disease. Central questions about ApoE as a target emphasize genetic isoform, total levels, and lipidation status, as each play key roles in amyloidosis. In this study, the leading investigators Liao and Li provide elegant data characterizing the therapeutic promise of a novel anti-human ApoE antibody (HAE-4). HAE-4 preferentially recognizes unlipidated and aggregated ApoE3 and ApoE4, which represent a small fraction of total ApoE but may be critical pathological players. Both intracerebroventricular and intraperitoneal administration of HAE-4 into APP transgenic mice significantly reduced Aβ plaque load and Aβ accumulation in the brain. HAE-4 also recognizes ApoE in amyloid plaques in living animals after direct application to the brain surface or peripheral administration into living animals. Efficacy in plaque reduction requires microglial activation via an Fcγ effector response, as administration of similar antibodies mutated in the Fc effector domain failed to clear plaques.
Importantly, HAE-4 does not significantly alter the levels of total ApoE in the brain or plasma, which is critically important because a reduction of peripheral ApoE is expected to increase the risk of cardiovascular disease. Whether HAE-4 may or may not share the cerebrovascular adverse effects that can be observed with anti-Aβ antibodies will be an important future question. Whether mice treated with HAE-4 have preserved cognitive function will also be important to address in the near future.
HAE-4 represents a very promising new approach to stimulate plaque removal, and may be particularly important for ApoE4 carriers, who form the majority of AD patients and for whom identification of potentially effective treatments has been particularly challenging.
Hong Kong University of Science & Technology
This very convincing study clearly showed that anti-ApoE antibodies selective for non-lipidated, aggregated ApoE can bind to ApoE in amyloid plaques and reduce Aβ accumulation. This effect is very similar to how anti-Aβ antibodies specific for aggregated Aβ reduce amyloid, such as Biogen's aducanumab, being tested in clinical trials for AD. As Aβ and ApoE are two major components of amyloid plaques, it would be interesting to test whether a combination of antibodies to both can have additive or synergistic effects on reducing or removing amyloid plaques. Because aggregation of ApoE, in particular ApoE4, likely accelerates AD-related pathologies in addition to Aβ, targeting ApoE can offer an opportunity to diversify strategies for AD therapy.
Indiana University School of Medicine
Liao et al. have reported that antibodies directed at human ApoE isoforms can reduce amyloid plaque burden in a mouse model of AD expressing human ApoE4. This study, like its predecessors from the Holtzman lab, derives from the recognition that plaques in the AD brain are formed through co-deposition of Aβ with insoluble forms of ApoE—which likely comprise a significant fraction of the mass of the plaque. The key finding from their previous work was that antibodies targeting murine ApoE had the unexpected effect of catalyzing plaque removal. The mechanism subserving this effect was postulated to be through microglial phagocytosis and this has been verified in the present study. They nicely showed that the loss of plaque was reliant upon Fc receptor binding.
The central outcome of the present study is that they can selectively target non-lipidated or poorly lipidated human ApoE species using newly generated antibodies. The antibody HAE-4 recognizes both non-lipidated human ApoE3 and 4 isoforms, which are preferentially associated with Aβ plaques. As a consequence, administration of HAE-4 did not affect the total levels of ApoE in either brain or plasma since essentially all ApoE exists in vivo incorporated into a lipidated HDL. This feature of the antibody has the virtue of extending the half-life of the antibody in plasma.
The ability to selectively target deposited, non-lipidated forms of ApoE as a means of lowering plaque burden is therapeutically attractive, given the importance of ApoE-mediated lipid transport in the brain. It is clear from the previous mouse studies that the window for any ApoE-based therapeutics is likely to be prior to plaque initiation. It is unknown whether HAE-4 will have an effect on pre-existing plaques. It is noteworthy that the effect of anti-ApoE antibodies appears to be largely restricted to fibrillary, rather than compact, plaques. All of this begs the question of whether this approach will be of utility in humans, given the ambiguity about the functional significance of plaque burden on cognition. Overall, this is a very nice study that explores novel therapeutic approaches.
Make a Comment
To make a comment you must login or register.