Plaques Age Glial Precursors, Stoking Inflammation
Quick Links
A new study adds evidence that Alzheimer’s pathology makes nearby cells senescent. Scientists led by Mark Mattson, National Institute on Aging, Baltimore, report in the April 1 Nature Neuroscience that in both people and animals, oligodendrocyte precursor cells (OPCs) surrounding Aβ plaques stop differentiating into myelin-repairing oligodendrocytes. Instead, they release inflammatory molecules into their environment and leave damaged axons bare of myelin. Drugs that clear senescent cells—known as senolytics—eliminated senescent OPCs and reduced neuroinflammation, microgliosis, and Aβ load in transgenic mouse models of AD, all the while improving their learning and memory. The results tap senolytic drugs as a potential therapy for Alzheimer’s disease.
- Oligodendroctye precursors help repair damaged myelin.
- Meeting Aβ plaques makes them senescent and inflammatory.
- Clearing those senescent cells improves cognition in AD mice.
“I’m intrigued that oligodendrocyte precursor cells appear to be an important player in Alzheimer’s,” said Li-Huei Tsai of the Massachusetts Institute of Technology, who was not involved in the study. “They’ve not been closely investigated in AD.”
“Together with other studies published recently, this study shows that senescent cells accumulate in age-related neurodegenerative diseases, like Alzheimer’s,” wrote Valery Krizhanovsky, Weizmann Institute of Science, Rehovot, Israel, to Alzforum. “Elimination of these cells holds a lot of promise as a potential future treatment.” Krizhanovsky also was not involved in the study.
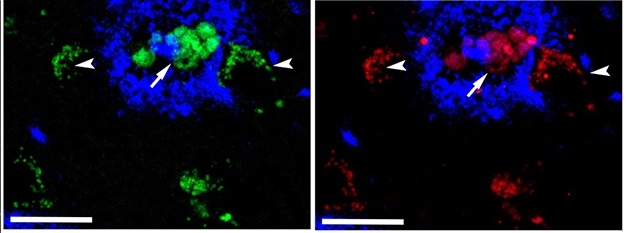
Plaques Evoke Senescence. When oligodendrocyte precursors (green) contact Aβ plaques (blue), they become senescent, expressing the tumor suppressor gene p21 (red). Arrows point to oligodendrocytes, arrowheads to their processes. [Courtesy of Zhang et al., Nature.]
Senescent cells are proliferative cells that have stopped dividing with age, usually after a certain number of divisions. They remain metabolically active, however, releasing proinflammatory cytokines. Senescent cells have been found to contribute to peripheral disorders, including diabetes, cancer, and atherosclerosis (Childs et al., 2016; Rodier and Campisi, 2011).
Scientists have started asking whether senescent cells accumulate in the brain. Miranda Orr, University of Texas Health, San Antonio, found that neurons containing tangles had entered a senescent state in both postmortem AD brain tissue and rTg4510 mice (Sep 2018 news). Darren Baker, Mayo Clinic, Rochester, Minnesota, reported that tau pathology caused senescence of astrocytes and microglia in PS19 mice, which express human mutant P301L tau. He did not examine OPCs. Both sets of researchers found that clearing away the aged cells prevented or slowed neurodegeneration and cognitive deficits in mice.
Do Aβ plaques bring about senescence in the brain? In the current study, first author Peisu Zhang and colleagues examined human postmortem tissue. In samples of the inferior parietal cortices of eight AD patients, eight with mild cognitive impairment, and eight age-matched controls, they used antibodies to label Aβ plaques, microglia, astrocytes, and OPCs. OPCs occur throughout the brain—even in gray matter where there are fewer myelinated axons than in white matter—and they migrate to sites of neurodegeneration to repair myelin there. In AD patients, OPCs co-localized with markers of senescence, namely tumor-suppressor proteins p16 and p21, in 80 percent of the plaques. Astrocytes and microglia did not appear to be senescent.
The authors saw the same pattern in the hippocampi of 4.5-month-old APPPS1 mice. OPCs—not microglia or astrocytes—were senescent around plaques. They expressed the inflammatory factors interleukin-1β and tumor necrosis factor-α and riled up microglia. Meanwhile, adjacent axons lost their myelin. These results suggested that plaques render OPCs senescent, which leads to inflammation, microgliosis, and denuded axons.
To test if Aβ alone could set this off, the researchers generated OPCs from mouse embryonic stem cells and exposed them to either varying concentrations of synthetic Aβ42 oligomers or vehicle. After seven days, none of the vehicle-treated cells became senescent, but up to 12 percent of the Aβ-treated cells did.
What if the researchers cleared senescent OPCs from mouse brains? Zhang treated APPPS1 mice with two FDA-approved senolytic compounds. Dasatinib and quercetin (D+Q) eliminate senescent cells from tissues by transiently inhibiting tyrosine kinases that suppress apoptosis, thus killing only senescent cells. Because it takes time for healthy, dividing OPCs to become senescent, the drugs can be given intermittently. In this way, treating 5-month-old APPPS1 mice for nine days halved OPC senescence. Microglia also assumed a less-activated appearance, marked by smaller cell bodies and branched processes. Once-weekly treatments for 11 weeks beginning at 3.5 months old almost eliminated senescent OPCs in the hippocampi of APPPS1 mice. These animals better remembered which arm they had previously explored in a Y maze and where the hidden platform was in a water maze. D+Q treated mice accumulated about one-third the Aβ plaque load and half the level of inflammatory cytokines in the hippocampus and entorhinal cortex, as untreated controls.
Mattson speculates that signaling pathways attract OPCs to sites of Aβ-induced myelin damage, and that they are stunned into senescence once they get there. Instead of replenishing oligodendrocytes and repairing the damage, they incite inflammation.
“This paper shows that senescent oligodendrocytes are a major contributor to the dysfunction that occurs in the brains of mice with neurodegeneration,” said James Kirkland, Mayo Clinic, Rochester, Minnesota, who has pioneered the use of senolytics in a range of diseases. “Targeting senescent cells could be a completely new way of going after neurodegenerative diseases like AD and Parkinson’s,” he said.
It’s early days for this strategy. One clinical trial has been completed so far on senolytics—a pilot for idiopathic pulmonary fibrosis, a fatal lung disease. The D+Q drug combination was deemed safe and seemed to benefit stamina and mobility. The trial continues open-label. Other trials are ongoing in age-related diseases, including frailty due to aging, diabetes, and kidney disease.
The new study adds OPCs to a growing list of cells—including microglia, astrocytes, and neurons—that can become senescent in neurodegenerative diseases. “It doesn’t seem to matter what the cell type is. Local accumulation of senescent cells is detrimental for overall function of the brain,” Baker told Alzforum. Orr concurred. “Collectively, our studies indicate that treatment with D+Q may be a useful approach to prevent or delay AD pathogenesis,” she wrote.—Gwyneth Dickey Zakaib
References
Research Models Citations
News Citations
Paper Citations
- Childs BG, Baker DJ, Wijshake T, Conover CA, Campisi J, van Deursen JM. Senescent intimal foam cells are deleterious at all stages of atherosclerosis. Science. 2016 Oct 28;354(6311):472-477. Epub 2016 Oct 27 PubMed.
- Rodier F, Campisi J. Four faces of cellular senescence. J Cell Biol. 2011 Feb 21;192(4):547-56. Epub 2011 Feb 14 PubMed.
External Citations
Further Reading
Papers
- Cui X, Guo YE, Fang JH, Shi CJ, Suo N, Zhang R, Xie X. Donepezil, a drug for Alzheimer's disease, promotes oligodendrocyte generation and remyelination. Acta Pharmacol Sin. 2019 Mar 27; PubMed.
- Hoch-Kraft P, Trotter J, Gonsior C. Missing in Action: Dysfunctional RNA Metabolism in Oligodendroglial Cells as a Contributor to Neurodegenerative Diseases?. Neurochem Res. 2019 Mar 6; PubMed.
- Cai Z, Xiao M. Oligodendrocytes and Alzheimer's disease. Int J Neurosci. 2015 Jul 14; PubMed.
Primary Papers
- Zhang P, Kishimoto Y, Grammatikakis I, Gottimukkala K, Cutler RG, Zhang S, Abdelmohsen K, Bohr VA, Misra Sen J, Gorospe M, Mattson MP. Senolytic therapy alleviates Aβ-associated oligodendrocyte progenitor cell senescence and cognitive deficits in an Alzheimer's disease model. Nat Neurosci. 2019 May;22(5):719-728. Epub 2019 Apr 1 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
UT Health San Antonio/South Texas VA
It is exciting to see continued interest surrounding cellular senescence in neurodegenerative diseases. Prior studies have reported senescent cells in postmortem human brains from patients with Alzheimer’s disease (Bhat et al., 2012; Musi et al., 2018), Parkinson’s disease (Chinta et al., 2018), and progressive supranuclear palsy (Musi et al., 2018). Mechanistically, tau accumulation induces cellular senescence in neurons (Musi et al., 2018), and microglia (Bussian et al., 2018). Zhang et al. now report that extracellular Aβ induces oligodendrocyte precursor cell (OPC) cellular senescence in Alzheimer’s disease. Their findings provide additional evidence that pathogenic protein accumulation induces cellular senescence in the brain, and that targeting cellular senescence may be an appealing therapeutic approach for clinical studies.
Extracellular Aβ and intracellular tau induce cell-type specific responses (both physiologically and pathogenically), which is highlighted by Zhang et al.’s findings. Their results indicate that in the plaque region, extracellular Aβ induces OPC cellular senescence but causes astrocyte and microglia activation. These results are also consistent with in vitro reports demonstrating OPC senescence in response to abnormal growth conditions/environments (Tang et. al., 2001). Future studies are needed to determine whether OPC cellular senescence is specific to Aβ, or whether other pathogenic protein aggregates (e.g., tau, α-synuclein, TDP-43, etc.) can induce a similar response. In our study we did not observe upregulated senescence-associated gene expression in 3xTgAD mice with high plaque burden without neurofibrillary tangles; however, we did not enrich for regions surrounding the plaques as done by Zhang et al. (Musi et al., 2018). For our analyses, we extracted RNA from whole mouse forebrain. Compared to age-matched controls, this methodology revealed a significant upregulation in senescence-associated gene expression in mice with neurofibrillary tangles, but not in mice with only plaque pathology. Collectively the studies suggest that tau pathology induces a greater senescent burden than Aβ plaque accumulation.
In general, neurodegenerative dementing disease research has not given OPCs and oligodendrocytes the same attention as other cell types. Investigating how the myelinating cells and their precursors respond to protein accumulation will continue to enhance our overall understanding of disease pathogenesis. For example, understanding the functional consequences of senescent OPCs will be an important next step in this line of investigation (i.e., can they migrate, respond to neuronal damage, differentiate, contribute to myelin repair, etc.?).
Zhang et al.’s work indicates that treating young mice with senolytics dasatinib and quercetin (D+Q) prevented Aβ accumulation and OPC senescence. It remains unknown whether the clearance of senescent OPCs without a reduction in Aβ (as shown in the acute nine-day study) is sufficient to improve behavior; or rather a reduction in Aβ is needed for these improvements (as shown with the 11-week treatment strategy). Also, the ability of D+Q to prevent plaque accumulation when given as a prophylactic treatment, prior to Aβ deposition and senescence onset, suggests “non-senolytic effects” of D+Q. Future studies will be needed to differentiate between senolytic and non-senolytic effects of D+Q and other drugs that target senescent cells.
Overall, it is promising to see prophylactic benefits of D+Q in a model of amyloidosis as it extends on our findings showing improvements using the same treatment in advanced neurodegeneration. Collectively our studies indicate that treatment with D+Q may be a useful approach to prevent or delay AD pathogenesis by reducing Aβ plaque accumulation and senescent OPCs as well as stop disease progression in advanced stages by reducing senescent neurofibrillary tangle-containing neurons.
References:
Bhat R, Crowe EP, Bitto A, Moh M, Katsetos CD, Garcia FU, Johnson FB, Trojanowski JQ, Sell C, Torres C. Astrocyte senescence as a component of Alzheimer's disease. PLoS One. 2012;7(9):e45069. PubMed.
Musi N, Valentine JM, Sickora KR, Baeuerle E, Thompson CS, Shen Q, Orr ME. Tau protein aggregation is associated with cellular senescence in the brain. Aging Cell. 2018 Dec;17(6):e12840. Epub 2018 Oct 11 PubMed.
Chinta SJ, Woods G, Demaria M, Rane A, Zou Y, McQuade A, Rajagopalan S, Limbad C, Madden DT, Campisi J, Andersen JK. Cellular Senescence Is Induced by the Environmental Neurotoxin Paraquat and Contributes to Neuropathology Linked to Parkinson's Disease. Cell Rep. 2018 Jan 23;22(4):930-940. Epub 2018 Jan 28 PubMed.
Bussian TJ, Aziz A, Meyer CF, Swenson BL, van Deursen JM, Baker DJ. Clearance of senescent glial cells prevents tau-dependent pathology and cognitive decline. Nature. 2018 Oct;562(7728):578-582. Epub 2018 Sep 19 PubMed.
Tang DG, Tokumoto YM, Apperly JA, Lloyd AC, Raff MC. Lack of replicative senescence in cultured rat oligodendrocyte precursor cells. Science. 2001 Feb 2;291(5505):868-71. Epub 2001 Jan 18 PubMed.
Make a Comment
To make a comment you must login or register.