Synaptojanin 1 Gene Affects Age of Onset in Familial AD, Memory in Mice
Quick Links
Variants in the gene for the lipid phosphatase synaptojanin 1 alter the age of onset in familial Alzheimer’s disease, says a new study. The work, from Catherine Marquer’s lab at Columbia University Medical Center in New York, also links overexpression of the gene to synapse loss in adults with Down’s syndrome/AD. In mice, elevation of synaptojanin 1 expression hypes up hippocampal neurons, to the detriment of memory. The study, which appeared June 5 in Cell Reports, raises the prospect that this brain-specific enzyme could offer a potential therapeutic target for memory impairment.
- Synaptojanin 1 variants reported to affect age of onset in FAD.
- High levels of this lipid phosphatase come with low synaptophysin in adults with Down’s syndrome.
- Overexpression in mice causes hippocampal excitability, memory problems.
In neurons, synaptojanin 1 catalyzes the dephosphorylation of membrane phosphoinositides. This is important for regulating synaptic vesicle recycling after neurotransmitter release. Its expression is elevated in two groups of people at risk for AD—carriers of the ApoE4 allele (Zhu et al., 2015), and people with Down’s syndrome (Martin et al., 2014). In animal models of AD, reducing synaptojanin 1 levels improves pathological measures and behavior (Zhu et al., 2013; McIntire et al., 2012; Apr 2008 news).
In the new work, first authors Andre Miranda, Mathieu Herman, and Rong Cheng offer genetic evidence to implicate synaptojanin 1 in additional, familial forms of AD. They identified variants associated with age of onset or memory function in a family with early Alzheimer disease due to the PSEN1 G206A mutation. In a different cohort of late-onset familial AD, they associate two SYNJ1 haplotypes with age of onset. The variants are in regions likely to influence gene function or expression, the scientists claim.
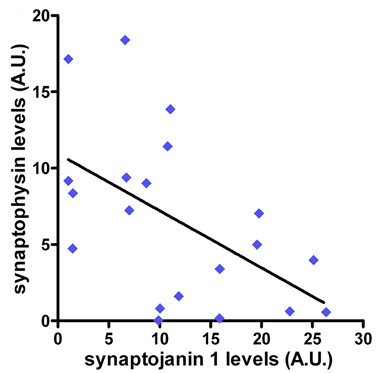
Synapses in Decline.
In postmortem brain tissue from adults with Down’s syndrome, higher synaptojanin 1 correlates with lower synaptophysin, indicating fewer intact synapses. [From Miranda et al., Cell Reports 2018.]
Examination of postmortem brain tissue from adults with Down’s syndrome revealed a potential link between synaptojanin 1 expression and synapse loss. The investigators found a modest inverse correlation between synaptojanin 1 levels and synaptophysin, a marker of synapse number—when synaptojanin 1 was high, synaptophysin, and presumably synapse number, dropped. The authors did not see the correlation in younger people with Down’s syndrome, in whom synaptojanin 1 levels were not yet elevated.
To directly test what excess synaptojanin 1 does to synaptic function, the scientists turned to a transgenic mouse overexpressing murine synj1. With a 76 percent surfeit of synaptojanin 1 protein compared to wild-type, the mice recapitulate the overexpression reported for ApoE4 carriers with early onset AD. The mice displayed no changes in synaptophysin or the postsynaptic marker PSD95, suggesting their synapses were intact. However, synaptic function was not. In two hippocampal-dependent memory tasks, the mice displayed age-related defects. In the radial arm water maze, young transgenic mice learned and remembered how to find a platform as well as the wild-type. However, as they got older, the transgenic mice seemed to forget the platform location from day to day when repeatedly tested. A fear conditioning test yielded similar results, where the transgenic mice seemed to forget the contextual cues they’d learned in the session before.
This may have been due to dysfunction of place cells in the hippocampus: When the investigators implanted electrodes in the hippocampi of mice and recorded neuronal firing in freely moving animals, the place cells in transgenics responded to locations less accurately and with more variability than those in wild-type animals. “Normally, we would see good repetition of firing at a specific location, but in the transgenic, the neurons fired at the correct location one day, but the next day they were not able to fire at the same location in the exact same way,” Marquer said. This could explain why the mice seemed a bit lost on their second day in the water maze, she said.
If increased synaptojanin 1 worsens memory, then inhibiting the enzyme could become a therapeutic strategy, the authors suggest. The right balance may be hard to strike, however, since loss-of-function mutations in synaptojanin 1 cause early onset Parkinson’s disease, seizures, and tau pathology (Dyment et al., 2015). “We want to inhibit the excess activity, but don’t want to go further than threshold level of normal function. We would need more of a regulator than complete inhibitor,” said Marquer.
William Mobley, University of California, San Diego, said the data support the role of synaptojanin 1 in synapse decline, but took a cautious view of its salience as a drug target. “It is one of several different genes that regulate endocytosis and should be looked at in the future in preclinical models. But one should not jump to the conclusion that this is the only, or the most important, cause of endosome dysfunction in AD,” he told Alzforum. The amyloid precursor protein, and its products, also regulate endosomes, and their role must be taken into account, Mobley said.—Pat McCaffrey
References
News Citations
Paper Citations
- Zhu L, Zhong M, Elder GA, Sano M, Holtzman DM, Gandy S, Cardozo C, Haroutunian V, Robakis NK, Cai D. Phospholipid dysregulation contributes to ApoE4-associated cognitive deficits in Alzheimer's disease pathogenesis. Proc Natl Acad Sci U S A. 2015 Sep 22;112(38):11965-70. Epub 2015 Sep 8 PubMed.
- Martin SB, Dowling AL, Lianekhammy J, Lott IT, Doran E, Murphy MP, Beckett TL, Schmitt FA, Head E. Synaptophysin and synaptojanin-1 in Down syndrome are differentially affected by Alzheimer's disease. J Alzheimers Dis. 2014;42(3):767-75. PubMed.
- Zhu L, Zhong M, Zhao J, Rhee H, Caesar I, Knight EM, Volpicelli-Daley L, Bustos V, Netzer W, Liu L, Lucast L, Ehrlich ME, Robakis NK, Gandy SE, Cai D. Reduction of synaptojanin 1 accelerates Aβ clearance and attenuates cognitive deterioration in an Alzheimer mouse model. J Biol Chem. 2013 Nov 1;288(44):32050-63. PubMed.
- McIntire LB, Berman DE, Myaeng J, Staniszewski A, Arancio O, Di Paolo G, Kim TW. Reduction of synaptojanin 1 ameliorates synaptic and behavioral impairments in a mouse model of Alzheimer's disease. J Neurosci. 2012 Oct 31;32(44):15271-6. PubMed.
- Dyment DA, Smith AC, Humphreys P, Schwartzentruber J, Beaulieu CL, FORGE Canada Consortium, Bulman DE, Majewski J, Woulfe J, Michaud J, Boycott KM. Homozygous nonsense mutation in SYNJ1 associated with intractable epilepsy and tau pathology. Neurobiol Aging. 2015 Feb;36(2):1222.e1-5. Epub 2014 Sep 6 PubMed.
Other Citations
Further Reading
No Available Further Reading
Primary Papers
- Miranda AM, Herman M, Cheng R, Nahmani E, Barrett G, Micevska E, Fontaine G, Potier MC, Head E, Schmitt FA, Lott IT, Jiménez-Velázquez IZ, Antonarakis SE, Di Paolo G, Lee JH, Hussaini SA, Marquer C. Excess Synaptojanin 1 Contributes to Place Cell Dysfunction and Memory Deficits in the Aging Hippocampus in Three Types of Alzheimer's Disease. Cell Rep. 2018 Jun 5;23(10):2967-2975. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
The University of Minnesota
This paper identified synaptojanin (synj1) variants associated with age of onset in both early and late-onset FAD cohorts. They also linked excess synj1 protein levels to synaptic loss and dysfunction in postmortem DS brains, as well as to hippocampus-based memory deficits in a mouse model overexpressing murine synj1.
These findings are very interesting. We have previously shown that increased synj1 expression contributes to ApoE4-associated cognitive deficits. The observations by Marquer and colleagues, in combination with ours (Zhu et al., 2015; Zhu et al., 2013) and others (McIntire et al., 2012) suggest that synaptojanin could be a good therapeutic target for treating cognitive impairment in AD.
For future studies, I would be interested in knowing how these synj1 SNPs affect synj1 function leading to AD-related pathology. Are they manifested as gain-of-toxic effects, which are different from synj1 variants with loss-of-function effects identified in PD patients?
I am also very intrigued by the results showing inverse correlation between synj1 protein expression and synaptic integrity in DS human brains. It would be interesting to see if similar patterns can be observed in postmortem AD brain tissue, and if ApoE4 worsens these changes based on our prior observations. Our previous data suggest that elevated synj1 expression in ApoE4 carriers was most prominent in early AD stages. It is possible that elevated synj1 expression with associated neuronal hyper-excitability is manifested as an early AD phenomenon which plays an important role in disease pathogenesis and development.
References:
Zhu L, Zhong M, Elder GA, Sano M, Holtzman DM, Gandy S, Cardozo C, Haroutunian V, Robakis NK, Cai D. Phospholipid dysregulation contributes to ApoE4-associated cognitive deficits in Alzheimer's disease pathogenesis. Proc Natl Acad Sci U S A. 2015 Sep 22;112(38):11965-70. Epub 2015 Sep 8 PubMed.
Zhu L, Zhong M, Zhao J, Rhee H, Caesar I, Knight EM, Volpicelli-Daley L, Bustos V, Netzer W, Liu L, Lucast L, Ehrlich ME, Robakis NK, Gandy SE, Cai D. Reduction of synaptojanin 1 accelerates Aβ clearance and attenuates cognitive deterioration in an Alzheimer mouse model. J Biol Chem. 2013 Nov 1;288(44):32050-63. PubMed.
McIntire LB, Berman DE, Myaeng J, Staniszewski A, Arancio O, Di Paolo G, Kim TW. Reduction of synaptojanin 1 ameliorates synaptic and behavioral impairments in a mouse model of Alzheimer's disease. J Neurosci. 2012 Oct 31;32(44):15271-6. PubMed.
Tel Aviv University
This study adds new evidence to the concept that dysregulation of endocytic recycling of synaptic vesicles in neurons is an important biological pathway in the etiology of disorders associated with neuronal hyper-excitability. The authors found that synj1 polymorphisms are associated with familial and sporadic Alzheimer’s disease. In animal studies, the authors show that AD-associated synj1 overexpression causes an increase in the firing rate of CA1 principal cells, destabilization and enlargement of place fields, and hippocampus-dependent cognitive impairments. These results point to a causal link between increased synj1 levels and impairments in functioning of CA1 excitatory neurons.
Interestingly, autosomal-recessive inherited synj1 variants, leading to the loss of synj1 phosphatase function, recently have been shown to cause early onset pharmaco-resistant seizures and progressive neurological decline (Hardies et al., 2016). Moreover, mouse knockout studies have shown that loss-of-function variants in endophilin and amphiphysin, two synj1 binding proteins, also lead to severe epilepsy (Di Paolo et al., 2002; Milosevic et al., 2011). Thus, the fact that reduction in synj1 levels causes epilepsy should be taken into account when developing AD therapeutic strategies based on synj1-mediated inhibition of synaptic vesicle endocytosis pathway. Increased understanding of the bell-shaped relationship between synj1 expression and neuronal activity, at the single-neuron and network levels, will guide future development of novel treatments for AD and epilepsy patients.
References:
Hardies K, Cai Y, Jardel C, Jansen AC, Cao M, May P, Djémié T, Hachon Le Camus C, Keymolen K, Deconinck T, Bhambhani V, Long C, Sajan SA, Helbig KL, AR working group of the EuroEPINOMICS RES Consortium, Suls A, Balling R, Helbig I, De Jonghe P, Depienne C, De Camilli P, Weckhuysen S. Loss of SYNJ1 dual phosphatase activity leads to early onset refractory seizures and progressive neurological decline. Brain. 2016 Sep;139(Pt 9):2420-30. Epub 2016 Jul 19 PubMed.
Di Paolo G, Sankaranarayanan S, Wenk MR, Daniell L, Perucco E, Caldarone BJ, Flavell R, Picciotto MR, Ryan TA, Cremona O, De Camilli P. Decreased synaptic vesicle recycling efficiency and cognitive deficits in amphiphysin 1 knockout mice. Neuron. 2002 Feb 28;33(5):789-804. PubMed.
Milosevic I, Giovedi S, Lou X, Raimondi A, Collesi C, Shen H, Paradise S, O'Toole E, Ferguson S, Cremona O, De Camilli P. Recruitment of endophilin to clathrin-coated pit necks is required for efficient vesicle uncoating after fission. Neuron. 2011 Nov 17;72(4):587-601. PubMed.
Make a Comment
To make a comment you must login or register.