Finally, a Diagnostic Marker for Lewy Body Disease?
Quick Links
Scientists are a step closer to a diagnostic test for Lewy body disease. At AAIC2023, held July 16 to 20 in Amsterdam, Oskar Hansson, Lund University, Sweden, reported that an α-synuclein-based seed amplification assay identified people with LBD among both cognitively impaired and unimpaired volunteers in the Swedish BioFinder cohort. Notably, about half the impaired people who tested positive for Lewy bodies also had amyloid pathology. Having Lewy pathology came with faster cognitive decline, regardless of amyloid.
- CSF assays identify people with Lewy body pathology.
- Lewy bodies speed progression in people with Alzheimer’s.
- The assays could help interpret clinical trial outcomes.
Within two years, Hansson hopes to use the test to help diagnose LBD in clinical practice. His team collaborated with Piero Parchi’s group at the University of Bologna, Italy. Their findings appeared back-to-back on July 18 in Nature Medicine.
Also at AAIC, Lawren Vandevrede, University of California, San Francisco, reported that a similar CSF assay distinguished autopsy-confirmed LBD from controls with high specificity and sensitivity, but only when the Lewy bodies were predominantly in the cortex and limbic areas of the person's brain. This assay worked less well among people who had Lewy bodies only in the amygdala or brainstem and who typically had milder symptoms.
Still, Vandevrede believes that, overall, seed amplification assays will prove useful for diagnosis. They perform better than relying on clinical symptoms, which are not always present or specific to LBD. Vandevrede and Hansson both emphasized that α-synuclein assays will be particularly important for running and interpreting drug studies. For example, they could explain why some people respond poorly to anti-amyloid removal.
“The quantification of LB pathology in vivo holds dual relevance. One is within a clinical framework, offering improved individualized prognostic insights, and the other in the context of clinical trials for all neurodegenerative disorders where a substantial number of cases are marked by the presence of LB pathology,” wrote Dag Aarsland, King’s College London, and Maria Camila Gonzalez Velez, Stavanger University Hospital, Norway.
Diagnosing DLB
Lewy bodies, those protein inclusions consisting primarily of α-synuclein fibrils, are found in several neurodegenerative diseases, including Parkinson’s, multiple system atrophy, and dementia with Lewy bodies (DLB). As many as half the people diagnosed with Alzheimer’s disease also have these deposits (Gibb et al., 1989; Hamilton, 2000; Jellinger, 2003). Indeed, across age-related dementias, Lewy bodies come second in prevalence only to amyloid plaques and tau tangles in AD.
Unlike plaques and tangles, Lewy bodies have been difficult to detect with fluid markers. Likewise, PET tracers for synucleinopathies are only just being developed (Part 5 of this series). With no good biomarkers, and symptoms that are easily confused with those of related disorders, diagnosing DLB is challenging, and the extent to which Lewy pathology contributes to cognitive decline in mixed pathologies remains undeciphered.
Enter seed amplification assays. For SAAs, scientists take a small sample of cerebrospinal fluid or tissue extract, incubate it with monomers of recombinant α-synuclein, and wait to see if aggregates form. They shake the solution to nudge seeds to nucleate and fibrils to grow, which can take days. In so doing, scientists led by Claudio Soto, University of Texas Medical School at Houston, detected α-synuclein fibrils in the CSF of PD patients seven years ago (Dec 2016 news). Since then, a flurry of SAA studies have reported synuclein seeds in the CSF of people with prodromal or fully fledged PD, culminating in a study of 1,100 people in the Parkinson’s Progression Markers Initiative that appeared last April (Rossi et al., 2020; Orru et al., 2021; Iranzo et al., 2021; April 2023 conference news).
Scientists found synuclein seeds in CSF from people with other LBDs, as well. Last year, Hansson and colleagues reported that, in BioFinder and a neuropathological cohort called the Arizona Study of Aging and Neurodegenerative Disorders, an α-synuclein SAA distinguished people with PD, Parkinson’s disease dementia, or DLB from controls with high sensitivity and specificity (Hall et al., 2022). As for mixed pathology, scientists led by Andrea Pilotto at the University of Brescia, Italy, found α-synuclein seeds in the CSF of 36 out of 80 people with AD (Pilotto et al., 2023). Now, Parchi and Hansson have correlated Lewy body with amyloid and tangle pathology, and estimated how the former changes clinical outcomes over time.
In Amsterdam, Hansson reported that among the 1,182 cognitively unimpaired people in BioFinder, 8 percent tested positive for Lewy body pathology, and its prevalence increased with age. About 60 percent were men, in keeping with their higher likelihood of getting LBD than women. Plaques or tangles were more prevalent, cropping up in 23 and 16 percent, respectively (image below). That said, 13 percent of those with plaques had LBD as well.
“I thought it extremely interesting that Lewy bodies were significantly more common in people who were amyloid-positive,” said Hansson. Co-first authors Sebastian Palmqvist from Lund and Marcello Rossi from the Istituto delle Scienze Neurologiche di Bologna (ISNB) calculated that having amyloid raised a person’s odds of developing Lewy bodies by 70 percent. Tangles had no correlation with Lewy bodies.
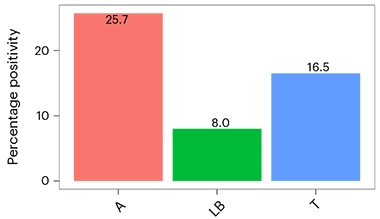

Lewy Body Effects. In BioFinder (top), half as many people had Lewy bodies (LB) as had neurofibrillary tangles (T). Having Lewy bodies accelerated disease progression, as seen by changes in global cognition (bottom left), memory (bottom middle), and attention/executive function (bottom right). [Courtesy of Palmqvist et al., 2023.]
Lewy body pathology mattered. People with it did worse on memory tests than controls with no AD or LB pathology. Their scores were on par with those of people who had tangles, a pathology that tracks with cognitive loss in AD. A positive LB test also correlated with loss of smell, a sign of impending LBD. Synuclein pathology seemed to accelerate AD because, over a period of 10 years, people with amyloid, tangles, and Lewy bodies slipped faster on measures of global cognition, memory, and attention/executive function than did people with either AD or LB pathology alone.
The seeding assay was also prognostic for symptomatic LBD. Some who tested positive subsequently developed DLB or PD; nobody who was negative did. “The average follow-up was 4.2 years, so we will need to see what happens over a longer time frame,” said Hansson.
What about people who were already symptomatic? In the second paper, co-first authors Corinne Quadalti at ISNB and Palmqvist reported similar effects. Of 883 BioFinder participants with mild cognitive impairment or dementia, 204, or 23 percent, had LB pathology; half of them also had plaques and tangles.
The prevalence of LB pathology increased with age but, paradoxically, less so than in the cognitively normal group. The authors suspect that people with other pathologies, such as TDP-43 encephalopathy or vascular disease, might dilute the number of LBD cases in a memory clinic setting. As Palmqvist and colleagues had found for the cognitively unimpaired group, Lewy bodies accelerated decline in measures of cognition, attention/executive function, and memory in the symptomatic cohort (image below).
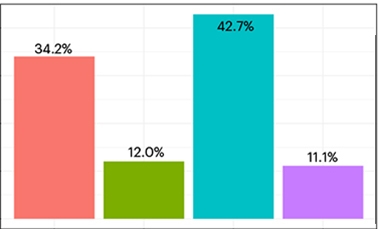

Faster Downhill. Nearly one in four symptomatic BioFinder participants have Lewy body pathology (top, green and purple). They deteriorate faster (bottom) in measures of (from let to right) global cognition, executive function, memory, and visuospatial function. [Image courtesy Quadalti et al., 2023.]
For the most part, the data confirm prior findings in neuropathological cohorts, namely that people with mixed AD/Lewy pathology got worse faster (Brenowitz et al., 2017; Malek-Ahmadi et al., 2019; Ryman et al., 2021; Gu et al., 2022).
The assay now allows clinicians to sharpen prognosis, said Hansson. “It opens up the possibility to study the clinical effects longitudinally, and that is quite helpful for clinical trials,” Hansson told Alzforum. “It is important to better understand variability in response to trajectories and therapies to see what happens to these individuals.”
Doug Galasko, University of California, San Diego, agreed. “With the approval of amyloid immunotherapy for AD, the effects and benefits of this treatment in patients who have AD plus positive SAA should be formally studied,” he wrote (comment below). “It will be of interest to see whether anti-amyloid antibodies have weaker clinical effects in such patients, or whether indeed their use attenuates synuclein pathology.”
In one case study, Vandevrede reported clinical outcomes for a 60-year-old woman whose memory had been sliding for five years, and who received 10 mg/Kg aducanumab in the Engage trial. Over six months, the treatment cleared some amyloid, 60 to 51 centiloids, yet her MMSE fell from 24 to 19. Eight months later, she was at 14, and dropped out of the trial. During the next three years, she increasingly showed symptoms typical of DLB, including Parkinsonism, paranoia, anxiety, and profound dysautonomia, or loss of regulatory control over autonomic nervous system function, such as heart rate, breathing, and swallowing. When she died at age 64, she was found to have high levels of plaques and tangles and neocortical Lewy body pathology. “I’m not suggesting we don’t include such patients based on this one case, but it does raise the question of what is the response of patients with co-pathology if you treat with amyloid-lowering or -targeting treatments,” said Vandevrede. “Using some of the tools we now have available for CSF will help us answer that question.”
Galasko noted that while SAAs may pave the way for prevention studies, he wants a less-invasive approach than CSF sampling for screening or enriching populations. Scientists in Japan led by Nobutaka Hattori at Juntendo University School of Medicine, Tokyo, recently reported that they detect α-synuclein seeds in serum. Their amplification assay distinguished people with PD and DLB from controls with accuracies of 96 and 90 percent, respectively (Okuzumi et al., 2023). “We don’t yet have a very reliable blood test, but that might just be a matter of time,” said Hansson.
Clinical Diagnosis Versus Biomarkers
Vandevrede’s data suggests that seed amplification assays can identify limbic and neocortical LBD better than clinical diagnosis. He compared the SYNTap α-synuclein seeding amplification test sold by Amprion, San Diego, against autopsy and clinical data from a small cohort of 56 donors at the Memory and Aging Center at UCSF. Of these, nine had predominantly limbic/neocortical LBD, six had brainstem pathology, and 14 had it in their amygdala. Neurologists consider limbic/neocortical LBD to be the full phenotype, since this is typically accompanied by symptoms of DLB and/or PD. What these brainstem- and amygdala-predominant forms mean clinically is unclear, said Vandevrede, though he suspects they represent prodromal stages.
Overall, SYNTap of CSF identified LBD with 96 percent specificity and 59 percent sensitivity. For those who had limbic/neocortical pathology, those numbers were 96 and 100 percent, respectively. For the amygdala and brainstem categories, sensitivities were only 43 and 17 percent, respectively. Hansson and colleagues had found similar sensitivities in the BioFinder cohort. The data suggest that the prodromal forms are releasing far fewer α-synuclein seeds into the CSF. Indeed, people with brainstem/amygdala LBs who did test positive also hallucinated or had REM sleep disorder, suggesting they may be on their way to more severe disease. “SYNTap may be identifying ‘seed-competent’ subgroups with clinical relevance,” said Vendevrede.
Can seed amplification assays help if they miss most cases? “The amygdala-only variant contributes to a substantial proportion of AD cases with α-synuclein pathology in postmortem studies and would thus go largely unnoticed,” noted Hansson and colleagues. “However, the amygdala-predominant variant does not lead to a clinical LBD presentation and is less clinically relevant,” they wrote. The implication is that the seeding assays will identify any LB pathology that is likely to contribute to clinical decline, including among people with AD co-pathology.
As for limbic/neocortical LBD, the assay's near-perfect performance in this small cohort suggests it may outperform clinical diagnosis, said Vandevrede. While everyone in this group was positive on SYNTap, 44 percent had hallucinations, only 22 percent REM sleep disorder, suggesting the assay picks up even mild disease.
On the other side of the coin, SAAs, such as real-time quaking induced conversion, or RT-QuIC, might correct clinical misdiagnoses. Galasko and colleagues tested CSF of about 200 clinically diagnosed DLB cases. Almost 30 percent tested negative in the seeding assay, suggesting that typical DLB symptoms, such as parkinsonism, hallucinations, and REM sleep disorder, occurring in non-DLB disorders might muddy the waters. “The bottom line is that people can overdiagnose DLB based on the clinical criteria, assuming that CSF RT-QuIC is ground truth,” wrote Galasko. Palmqvist was surprised by that interpretation. “Our data show that we often miss LB pathology,” he wrote. Galasko noted that the cohorts and how they are referred to memory centers are different. “When people are being recruited for a DLB study, we need to be careful about mimics—a little Parkinsonism or disrupted sleep that sounds like RBD can drive false positives,” he wrote.—Tom Fagan
References
News Citations
- Spying on α-Synuclein Inclusions: PET Tracers Inch Closer to Success
- Methods to Detect Amyloid Seeds Improve, Extend to Blood and Parkinson’s
- Synuclein Assay Passes the Sniff Test—What of Other Seeds?
Paper Citations
- Gibb WR, Mountjoy CQ, Mann DM, Lees AJ. A pathological study of the association between Lewy body disease and Alzheimer's disease. J Neurol Neurosurg Psychiatry. 1989 Jun;52(6):701-8. PubMed.
- Hamilton RL. Lewy bodies in Alzheimer's disease: a neuropathological review of 145 cases using alpha-synuclein immunohistochemistry. Brain Pathol. 2000 Jul;10(3):378-84. PubMed.
- Jellinger KA. Alpha-synuclein pathology in Parkinson's and Alzheimer's disease brain: incidence and topographic distribution--a pilot study. Acta Neuropathol. 2003 Sep;106(3):191-201. PubMed.
- Rossi M, Candelise N, Baiardi S, Capellari S, Giannini G, Orrù CD, Antelmi E, Mammana A, Hughson AG, Calandra-Buonaura G, Ladogana A, Plazzi G, Cortelli P, Caughey B, Parchi P. Ultrasensitive RT-QuIC assay with high sensitivity and specificity for Lewy body-associated synucleinopathies. Acta Neuropathol. 2020 Jul;140(1):49-62. Epub 2020 Apr 27 PubMed. Correction.
- Orrù CD, Ma TC, Hughson AG, Groveman BR, Srivastava A, Galasko D, Angers R, Downey P, Crawford K, Hutten SJ, Kang UJ, Caughey B. A rapid α-synuclein seed assay of Parkinson's disease CSF panel shows high diagnostic accuracy. Ann Clin Transl Neurol. 2021 Feb;8(2):374-384. Epub 2020 Dec 29 PubMed.
- Iranzo A, Fairfoul G, Ayudhaya AC, Serradell M, Gelpi E, Vilaseca I, Sanchez-Valle R, Gaig C, Santamaria J, Tolosa E, Riha RL, Green AJ. Detection of α-synuclein in CSF by RT-QuIC in patients with isolated rapid-eye-movement sleep behaviour disorder: a longitudinal observational study. Lancet Neurol. 2021 Mar;20(3):203-212. PubMed.
- Hall S, Orrù CD, Serrano GE, Galasko D, Hughson AG, Groveman BR, Adler CH, Beach TG, Caughey B, Hansson O. Performance of αSynuclein RT-QuIC in relation to neuropathological staging of Lewy body disease. Acta Neuropathol Commun. 2022 Jun 22;10(1):90. PubMed.
- Pilotto A, Bongianni M, Tirloni C, Galli A, Padovani A, Zanusso G. CSF alpha-synuclein aggregates by seed amplification and clinical presentation of AD. Alzheimers Dement. 2023 Aug;19(8):3754-3759. Epub 2023 Apr 27 PubMed.
- Brenowitz WD, Hubbard RA, Keene CD, Hawes SE, Longstreth WT Jr, Woltjer RL, Kukull WA. Mixed neuropathologies and associations with domain-specific cognitive decline. Neurology. 2017 Oct 24;89(17):1773-1781. Epub 2017 Sep 22 PubMed.
- Malek-Ahmadi M, Beach TG, Zamrini E, Adler CH, Sabbagh MN, Shill HA, Jacobson SA, Belden CM, Caselli RJ, Woodruff BK, Rapscak SZ, Ahern GL, Shi J, Caviness JN, Driver-Dunckley E, Mehta SH, Shprecher DR, Spann BM, Tariot P, Davis KJ, Long KE, Nicholson LR, Intorcia A, Glass MJ, Walker JE, Callan M, Curry J, Cutler B, Oliver J, Arce R, Walker DG, Lue LF, Serrano GE, Sue LI, Chen K, Reiman EM. Faster cognitive decline in dementia due to Alzheimer disease with clinically undiagnosed Lewy body disease. PLoS One. 2019;14(6):e0217566. Epub 2019 Jun 25 PubMed.
- Ryman SG, Yutsis M, Tian L, Henderson VW, Montine TJ, Salmon DP, Galasko D, Poston KL. Cognition at Each Stage of Lewy Body Disease with Co-occurring Alzheimer's Disease Pathology. J Alzheimers Dis. 2021;80(3):1243-1256. PubMed.
- Gu Y, Kociolek A, Fernandez KK, Cosentino SA, Zhu CW, Jin Z, Leverenz JB, Stern YB. Clinical Trajectories at the End of Life in Autopsy-Confirmed Dementia Patients With Alzheimer Disease and Lewy Bodies Pathologies. Neurology. 2022 May 24;98(21):e2140-e2149. Epub 2022 Apr 4 PubMed.
- Okuzumi A, Hatano T, Matsumoto G, Nojiri S, Ueno SI, Imamichi-Tatano Y, Kimura H, Kakuta S, Kondo A, Fukuhara T, Li Y, Funayama M, Saiki S, Taniguchi D, Tsunemi T, McIntyre D, Gérardy JJ, Mittelbronn M, Kruger R, Uchiyama Y, Nukina N, Hattori N. Propagative α-synuclein seeds as serum biomarkers for synucleinopathies. Nat Med. 2023 Jun;29(6):1448-1455. Epub 2023 May 29 PubMed. Correction.
Further Reading
Papers
- Kluge A, Bunk J, Schaeffer E, Drobny A, Xiang W, Knacke H, Bub S, Lückstädt W, Arnold P, Lucius R, Berg D, Zunke F. Detection of neuron-derived pathological α-synuclein in blood. Brain. 2022 Sep 14;145(9):3058-3071. PubMed. Correction.
Primary Papers
- Palmqvist S, Rossi M, Hall S, Quadalti C, Mattsson-Carlgren N, Dellavalle S, Tideman P, Pereira JB, Nilsson MH, Mammana A, Janelidze S, Baiardi S, Stomrud E, Parchi P, Hansson O. Cognitive effects of Lewy body pathology in clinically unimpaired individuals. Nat Med. 2023 Aug;29(8):1971-1978. Epub 2023 Jul 18 PubMed.
- Quadalti C, Palmqvist S, Hall S, Rossi M, Mammana A, Janelidze S, Dellavalle S, Mattsson-Carlgren N, Baiardi S, Stomrud E, Hansson O, Parchi P. Clinical effects of Lewy body pathology in cognitively impaired individuals. Nat Med. 2023 Aug;29(8):1964-1970. Epub 2023 Jul 18 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of California, San Diego
The remarkable ability of seed amplification assays (SAA), such as RT-QuIC, to detect pathological α-synuclein in CSF has enabled many interesting clinical questions to be addressed, in particular, those related to early diagnosis of synuclein disorders. The pair of recent papers from the BioFINDER cohort applied RT-QuIC to a large set of CSF samples. In cognitively unimpaired people (Palmqvist et al.) about 8 percent were positive, with greater likelihood related to age, male sex, positive AD (amyloid biomarkers) and, notably, olfactory loss. Long-term follow-up data showed a striking increased risk for RT-QuIC positive people to develop a clinical synucleinopathy, such as Parkinson’s Disease or DLB, as well as a risk for worsening of cognition.
The study of cognitively impaired people suggests that SAAs can be used to identify people at risk for synucleinopathies, as is being implemented at scale by the Parkinson’s Progression Markers Initiative. This may pave the way for prevention studies but we will need a less-invasive approach than CSF sampling for screening or enriching populations. The recent study of SAA using serum (Okuzumi et al., 2023) suggests that blood screening may become feasible in future. Combining SAA with olfactory screening may be a useful way to identify people at risk.
In people with MCI or mild dementia (Quadalti et al.), more than 20 percent had positive RT-QUiC tests, with a similar increased risk in people with amyloid co-pathology. The clinical phenotype at baseline did not always indicate DLB—sometimes hallucinations were present, although Parkinsonism was not assessed using a structured examination—but people with positive RT-QuIC had faster progression on tests of executive and visuospatial abilities. In symptomatic MCI or mild dementia, the rate of cognitive decline was faster in people who were SAA positive, particularly for the combination of A+S+. These findings support RT-QuIC as a way to identify DLB early in its clinical course, and extend many prior studies showing that people with AD and concomitant LB pathology progress faster.
With the approval of amyloid immunotherapy for AD, the effects and benefits of this treatment in patients who have AD plus positive SAA should be formally studied; it will be of interest to see whether anti-amyloid antibodies have weaker clinical effects in such patients, or whether their use attenuates synuclein pathology. The Palmqvist and Quadalti papers further the concept of using a panel of biomarkers to identify specific brain pathology to improve personalized predictions of clinical progression.
References:
Okuzumi A, Hatano T, Matsumoto G, Nojiri S, Ueno SI, Imamichi-Tatano Y, Kimura H, Kakuta S, Kondo A, Fukuhara T, Li Y, Funayama M, Saiki S, Taniguchi D, Tsunemi T, McIntyre D, Gérardy JJ, Mittelbronn M, Kruger R, Uchiyama Y, Nukina N, Hattori N. Propagative α-synuclein seeds as serum biomarkers for synucleinopathies. Nat Med. 2023 Jun;29(6):1448-1455. Epub 2023 May 29 PubMed. Correction.
King's College London
Stavanger University Hospital
For decades, the link between Lewy body (LB) pathology with parkinsonism and cognitive impairment has been known. Most commonly LB pathology clinically manifests as Parkinson’s disease dementia or dementia with Lewy bodies. Subsequent studies have highlighted the heightened clinical aggressiveness and accelerated decline observed in individuals whose brains exhibit both LB pathology and Alzheimer's disease pathology. This stands in contrast to those with singular pathologies, be it pure Alzheimer's (AD) or pure Lewy bodies. This evidence has largely been based on postmortem autopsy studies and preclinical animal data. Excitingly, contemporary advances now empower us to assess LB pathology in vivo through α-synuclein-seed amplification assays, such as real-time, quaking-induced conversion assay (RT-QuIC). Several recent studies from different laboratories have shown the very high sensitivity and specificity of RT-QUIC for PD and DLB, including prodromal DLB.
In the Swedish BioFinder study, a large cohort of people, with and without cognitive impairment, has provided CSF and other biomarkers and is being followed longitudinally. Palmqvist et al. demonstrated that individuals categorized as AD–/LB+ or AD+/LB– exhibited notably inferior performance in baseline global cognition and memory compared to their AD–/LB– counterparts.
Similarly, the study conducted by Quadalti et al. yielded significant insights. It revealed that while Aβ and tau showed no direct correlation, LB pathology displayed a robust association with hallucinations, as well as impaired attention, executive function, visuospatial skills, and motor abilities. Notably, LB pathology emerged as a pivotal factor in accelerating longitudinal decline, autonomously impacting this decline trajectory without being contingent on Aβ or tau.
The quantification of LB pathology in vivo holds dual relevance—one within a clinical framework, offering improved individualized prognostic insights, and the other in the context of clinical trials for all neurodegenerative disorders, where a substantial number of cases are marked by the presence of LB pathology. Together, these studies underline the importance of α-synuclein pathology also outside of typical Lewy body disease, and suggest that the novel technology might become a relevant screening test in memory clinics together with the traditional AD biomarkers.
University of Pennsylvania, Veterans Affairs Medical Center
Intraneuronal aggregates of α-synuclein (αSyn) are the hallmark of pathology in Lewy body diseases (LBD), including Parkinson’s disease (PD) and dementia with Lewy bodies (DLB) (Henderson et al., 2019). Diffuse cortical spread of αSyn pathology correlates strongly with cognitive decline in LBD with PD dementia (PDD) and in DLB, being nearly indistinguishable at autopsy (Kövari et al., 2003; Hurtig et al., 2000). There is a significant population of normal and cognitively impaired individuals who at autopsy have concurrent AD and LBD pathology. Over 25 percent of these individuals have primary LBD pathology meeting criteria for AD neuropathologic diagnosis, and up to 60 percent of them have primary AD pathology with concomitant LBD neuropathology (Hepp et al., 2016; Irwin et al., 2012). However, the interplay between Alzheimer’s and LBD pathology has been difficult to study in living individuals until the advent of seed aggregation assays (SAAs) as a biomarker for αSyn pathology. These studies from Palmqvist et al. and Quadalti et al. use novel αSyn SAAs to characterize cognitive trajectories as they relate to underlying AD and LBD pathology in cognitively unimpaired individuals and in those with mild impairment (Quadalti et al., 2023).
First, these studies reinforce the utility of aSyn SAA as a biomarker, given that only SAA+ individuals progressed to clinical LBD and their significant association with hyposmia, a known prodromal symptom in LBD. Notably, SAA-positivity was a better predictor of conversion to clinical LBD than a smell test. Second, the presence of LB pathology was associated with worse baseline cognition and memory and with worse longitudinal cognitive decline, implicating both an independent and synergistic effect with AD pathology. Interestingly, in the populations with cognitive impairment, people in the AD+LBD- biomarker group had a higher proportion of progression to a clinical diagnosis (50 percent to 93 percent AD, 0.5 to 0.8 percent DLB/PD) relative to those in the AD-LBD+ (8 percent to 14 percent AD, 31 percent to 46 percent DLB/PD) or AD+LBD+ (46 percent to 75 percent AD, 9 percent to 16 percent DLB/PD) groups. Whether these findings are due to length of follow-up (mean of four years in these studies), due to diagnosis setting (memory clinic rather than movement disorders clinic), or other factors remains to be seen. The heterogeneity of clinical diagnosis in the AD+LBD+ cohort suggests pathways to a diversity of clinical phenotypes.
The availability and clinical use of biomarkers for amyloid, tau, and αSyn pathology provides the opportunity to understand the interplay between these pathologies that contribute to cognitive decline and neurodegeneration. These studies are novel and exciting because for the first time they characterize early and prodromal populations, using underlying biomarkers, to begin to understand in vivo how LBD pathology interacts with AD pathology as well as to demonstrate the potential for selecting relevant individuals for clinical trials based on underlying pathology. Furthermore, prognostic information based on biomarker data may be useful for individuals who are known to be at high-risk for development of cognitive decline or neurodegeneration, especially with the idea of potentially testing drugs in those with early or prodromal disease. Future studies will need to enroll larger cohorts (given relative paucity of AD and LBD co-pathology) and have longer follow-up to fully understand the synergy of LBD and AD pathology.
References:
Henderson MX, Trojanowski JQ, Lee VM. α-Synuclein pathology in Parkinson's disease and related α-synucleinopathies. Neurosci Lett. 2019 Sep 14;709:134316. Epub 2019 Jun 3 PubMed.
Kövari E, Gold G, Herrmann FR, Canuto A, Hof PR, Bouras C, Giannakopoulos P. Lewy body densities in the entorhinal and anterior cingulate cortex predict cognitive deficits in Parkinson's disease. Acta Neuropathol. 2003 Jul;106(1):83-8. PubMed.
Hurtig HI, Trojanowski JQ, Galvin J, Ewbank D, Schmidt ML, Lee VM, Clark CM, Glosser G, Stern MB, Gollomp SM, Arnold SE. Alpha-synuclein cortical Lewy bodies correlate with dementia in Parkinson's disease. Neurology. 2000 May 23;54(10):1916-21. PubMed.
Hepp DH, Vergoossen DL, Huisman E, Lemstra AW, Netherlands Brain Bank, Berendse HW, Rozemuller AJ, Foncke EM, van de Berg WD. Distribution and Load of Amyloid-β Pathology in Parkinson Disease and Dementia with Lewy Bodies. J Neuropathol Exp Neurol. 2016 Oct;75(10):936-945. Epub 2016 Aug 11 PubMed.
Irwin DJ, White MT, Toledo JB, Xie SX, Robinson JL, Van Deerlin V, Lee VM, Leverenz JB, Montine TJ, Duda JE, Hurtig HI, Trojanowski JQ. Neuropathologic substrates of Parkinson disease dementia. Ann Neurol. 2012 Oct;72(4):587-98. PubMed.
Quadalti C, Palmqvist S, Hall S, Rossi M, Mammana A, Janelidze S, Dellavalle S, Mattsson-Carlgren N, Baiardi S, Stomrud E, Hansson O, Parchi P. Clinical effects of Lewy body pathology in cognitively impaired individuals. Nat Med. 2023 Aug;29(8):1964-1970. Epub 2023 Jul 18 PubMed.
Make a Comment
To make a comment you must login or register.