New PET Tracer Tracks Preclinical Movement Disorders
Quick Links
After years of searching for diagnostic biomarkers for neurodegenerative diseases, scientists more recently have begun focusing on markers that change as these disorders progress. Such markers could indicate if a treatment works. Two imaging markers that might track Huntington’s and Parkinson’s diseases even before symptoms set in stood out at the 2015 annual meeting of the American Academy of Neurology, held this year from April 18 to 25 in Washington, D.C. Both tracers bind predominantly in the striatum. The evidence is preliminary, but experts agreed it looks promising, especially for monitoring the earliest stages of both HD and PD.
Huntington’s disease takes a particular toll on the medium spiny neurons (MSNs) of the striatum. These neurons produce gobs of phosphodiesterase 10A, an enzyme involved in cell signaling. Because other neurons in the brain express PDE10A at lower levels and because it is one of the first proteins to wane in mouse models of HD, PDE10A makes an attractive biomarker candidate (see Hebb et al., 2004). The mutant huntingtin protein may also interact directly with the transcription machinery for PDE10A to reduce its expression (Hu et al., 2004). “The thought has been if we can track PDE10A expression over time, we will get a picture of MSN degeneration in the striatum,” said Sarah Janicki, Columbia University Medical Center, New York. “That may give a unique picture of brain changes in Huntington’s.”
At Molecular NeuroImaging (MNI) in New Haven, Connecticut, a privately held neuroimaging services company, a research group led by David Russell has developed an F18-labelled PET tracer for PDE10A called MNI-659. Data from humans and non-human primates suggests that the tracer specifically binds in brain areas known to express PDE10A, and in vitro assays reveal that MNI-659 competes for known inhibitors of PDE10A activity, Russell told Alzforum (see also Barret et al., 2014).
In a previous cross-sectional study, Russell found that in 11 carriers of the HD gene, loss of MNI-659 signal correlated both with the Unified Huntington’s Disease Rating Scale scores and the burden of pathology score. The latter score can estimate disease burden in mutant huntingtin carriers based on their CAG repeat length and age (see Russell et al., 2014). The MNI-659 signal dropped minimally in asymptomatic carriers, more in mildly symptomatic stage 1 patients, and still more in patients in stage 2 (see image below). This sample did not include people with moderate- to late-stage HD, i.e. stages 3 through 5. Two of the three people without symptoms registered at the low end of normal signal, while another had lost a bit of signal, suggesting that the tracer picks up change before clinical symptoms appear. “This might be one of the first biomarker indications of changes to come in HD,” Russell told Alzforum, perhaps even preceding striatal degeneration.
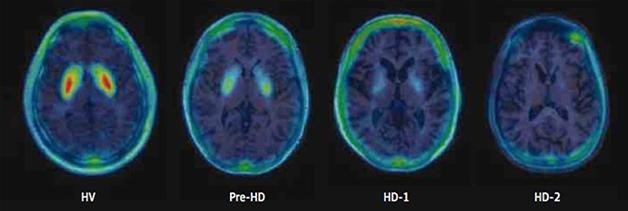
Steady Drop: MNI-659 signal falls gradually from healthy volunteers (HV) to people without symptoms (pre-HD) and early stages 1 and 2 HD. [© (2015) American Medical Association. All rights reserved.]
At AAN, Russell presented early longitudinal results on eight of those people. Two asymptomatic volunteers were five to 10 years from onset; three more people had stage 1 disease, and three stage 2. For all, the signal in the putamen and the caudate nucleus—the major divisions of the striatum—dropped over one year, regardless of the stage of disease. In the caudate, subjects averaged a 16.59 percent reduction per year, compared to 6.9 percent in the putamen and 5.81 percent in the globus pallidus. The rate of decline appears to continue, as the researchers have begun scanning these people for a second year of follow-up. So far, data on four participants suggest binding declines again by about 5 to 6 percent per year in the putamen and globus pallidus, and 15 to 20 percent per year in the caudate. At the earliest time points, lowered PDE10A expression likely accounts for most of the loss of signal, Russell said, while later on, dying neurons probably contribute.
To compare this rate of loss with healthy controls, Russell has taken single PET scans of a cohort of healthy controls. This cross-sectional data hints at an age-dependent loss of signal amounting to less than 1 percent per year in all striatal regions. That means the loss for HD patients speeds up at least five- to 15-fold, he said.
Given that MNI-659 binding declines early and differences are measurable within just a year, Russell believes it could make a good biomarker for striatal pathology in HD. It seems to start dropping in pre-symptomatic to early disease stages, meaning it may pick up on a crucial time window in HD, he added. The agent may be best suited for early disease, he told the audience, as binding appears to bottom out toward middle stages. One patient with stage 2 disease registered almost no signal, he pointed out.
MNI-659 could complement volumetric magnetic resonance imaging (MRI) and PET with fluorodeoxyglucose, which may be more informative later on, said Russell. A larger, longer, multi-site trial of MNI-659 with more patients representing a wider range of disease stages is needed.
Russell will test how this marker changes in Parkinson’s disease, or in other disorders that attack the striatum, such as schizophrenia or addiction.
Audience members asked know how MNI-659 PET related to MRI and FDG-PET. Russell said that he had no longitudinal MRI data, but previous cross-sectional study suggested that striatal volume decreased essentially in parallel with MNI-569 signal loss. However, MNI-659 seems to change first, as people with HD who still had normal striatal volume already registered MNI-659 loss. As for FDG-PET, Russell has no data on that marker to compare with MNI-659, but pointed out that it is known to mirror volume declines (see Tang et al., 2013). In Alzheimer’s disease and also frontotemporal dementia, the use of FDG-PET is declining as more data are becoming available for more disease-specific tracers of amyloid and tau pathology, respectively.
“These data looked really strong,” said session co-chair Victor Sung, University of Alabama School of Medicine, Birmingham. He asked how PDE10A inhibitors, which are being developed as a potential treatment for HD (Fusco and Giampà, 2015), would affect MNI-659 imaging. Russell said that since the drugs bind to the same region of PDE10A, they would likely displace the tracer.
Janicki agreed that the data were encouraging. “MNI-659 appears to be both sensitive and specific for the striatum,” she told Alzforum. She wants to know when signal starts to drop in pre-manifest patients relative to disease onset. “Hopefully this could be used to identify people most likely to convert [to symptomatic disease], so we might intervene as early as possible,” she said.
Parkinson’s and Beyond
Researchers at MNI are also hunting for imaging markers for PD progression. John Seibyl presented two-year follow-up data from 241 newly diagnosed patients in the Parkinson’s Progression Markers Initiative (PPMI) who underwent dopamine transporter (DaT) SPECT scans with 123-I-Ioflupane. This older tracer measures levels of the dopamine transporter. Researchers focus on signal loss in the striatum, as they do for MNI-659. However, whereas MNI-659 binds PDE10A produced in postsynaptic terminals of the striatum, the dopamine transporter appears on presynaptic terminals of axons that originate in the substantia nigra. A deteriorating 123-I-Ioflupane signal indicates a loss of those dopamine neurons.
Seibyl and colleagues examined change in the striatum as a whole, and in its component regions, i.e. the caudate, anterior putamen, and posterior putamen. They compared brain regions contralateral of the side of the body most affected by PD symptoms with brain regions on the ipsilateral side, which would be presumed to have less pathology (see image below).
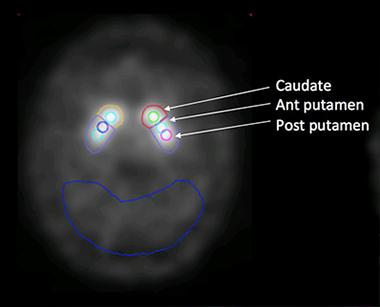
Regions of Interest:
The researchers look at tracer uptake in both sides of the striatum. The cerebellum (outlined in blue) serves as the reference. [Image courtesy of John Seibyl.]
In all patients, the contralateral (i.e., the more affected) side took up less tracer at baseline than the ipsilateral side. A year later, uptake in the contralateral side had fallen another 7.4 percent, and 15 percent by year two. Though the less affected side retained more tracer at baseline, it too lost binding capacity—11.8 percent in the first year, and 17.1 percent total over two years. This suggests that in newly diagnosed patients, the striatum connected to the less-affected half of the body loses relatively more DaT binding than the contralateral side. The biggest decrease seems to happen early in the disease, slowing down later on.
Analyzing each striatal region individually, the researchers found that those on the ipsilateral side followed a consistent pattern of signal loss: The more posterior the region—the posterior putamen, followed by the anterior putamen, and then the caudate—the greater the loss. The pattern on the contralateral side was less clear. There, the posterior putamen lost the least signal, compared with the other striatal regions. Seibyl said he is unsure why that would be.
How does the reduction in binding correlate with disease? Change in DaT scan signal did not correlate with decline on the Unified Parkinson’s Disease Rating Scale. This is not unusual, Seibyl told Alzforum, as biomarkers and clinical motor ratings measure different aspects of neural function.
The results hint that DaT scans could be a useful marker of early PD progression, but more work is required to figure out what a changing signal means, Seibyl said. Already, it appears as though researchers should consider striatal regions separately. “If you were designing a clinical trial of a putative neuroprotective compound, where in the striatum you sample may be important for determining the effect,” Seibyl said. Russell agreed. “The most prognostic information at baseline may be in the contralateral putamen, but to follow disease progression from there, the caudate or the ipsilateral putamen may be better since they won't ‘bottom out.’”
Just as in HD and PD, data are trickling in on molecular imaging in AD, particularly tau pathology. Brad Dickerson, Massachusetts General Hospital, Boston, presented data on the PET imaging ligand T807, much of which Alzforum covered at the Human Amyloid Imaging conference earlier this year (see Feb 2015 news). Dickerson presented one-year follow-up data on a person with frontotemporal dementia. He reported a tight correlation between the T807 binding pattern at baseline and at 12 months, suggesting good test-retest reliability. Binding increased by as much as 10 percent in some areas, hinting that the scan can detect accumulation over time. In addition, some areas that were bare at baseline but lay near affected regions—such as the medial temporal gyrus and supramarginal gyrus— took up tracer one year later, suggesting that the pathology had spread. From the perspective of tracking treatment, that is encouraging, suggested Dickerson. “If we had a disease-modifying therapy, we might be able to show efficacy if we detect reduction in that increase over time,” he said.—Gwyneth Dickey Zakaib
References
News Citations
Paper Citations
- Hebb AL, Robertson HA, Denovan-Wright EM. Striatal phosphodiesterase mRNA and protein levels are reduced in Huntington's disease transgenic mice prior to the onset of motor symptoms. Neuroscience. 2004;123(4):967-81. PubMed.
- Hu H, McCaw EA, Hebb AL, Gomez GT, Denovan-Wright EM. Mutant huntingtin affects the rate of transcription of striatum-specific isoforms of phosphodiesterase 10A. Eur J Neurosci. 2004 Dec;20(12):3351-63. PubMed.
- Barret O, Thomae D, Tavares A, Alagille D, Papin C, Waterhouse R, McCarthy T, Jennings D, Marek K, Russell D, Seibyl J, Tamagnan G. In Vivo Assessment and Dosimetry of 2 Novel PDE10A PET Radiotracers in Humans: 18F-MNI-659 and 18F-MNI-654. J Nucl Med. 2014 Aug;55(8):1297-304. Epub 2014 Jun 4 PubMed.
- Russell DS, Barret O, Jennings DL, Friedman JH, Tamagnan GD, Thomae D, Alagille D, Morley TJ, Papin C, Papapetropoulos S, Waterhouse RN, Seibyl JP, Marek KL. The phosphodiesterase 10 positron emission tomography tracer, [18F]MNI-659, as a novel biomarker for early Huntington disease. JAMA Neurol. 2014 Dec;71(12):1520-8. PubMed.
- Tang CC, Feigin A, Ma Y, Habeck C, Paulsen JS, Leenders KL, Teune LK, van Oostrom JC, Guttman M, Dhawan V, Eidelberg D. Metabolic network as a progression biomarker of premanifest Huntington's disease. J Clin Invest. 2013 Sep 3;123(9):4076-88. PubMed.
- Fusco FR, Giampà C. Phosphodiesterases as therapeutic targets for Huntington's disease. Curr Pharm Des. 2015;21(3):365-77. PubMed.
External Citations
Further Reading
Papers
- Dogan I, Eickhoff CR, Fox PT, Laird AR, Schulz JB, Eickhoff SB, Reetz K. Functional connectivity modeling of consistent cortico-striatal degeneration in Huntington's disease. Neuroimage Clin. 2015;7:640-52. Epub 2015 Feb 27 PubMed.
- Wassef SN, Wemmie J, Johnson CP, Johnson H, Paulsen JS, Long JD, Magnotta VA. T1ρ imaging in premanifest Huntington disease reveals changes associated with disease progression. Mov Disord. 2015 Jul;30(8):1107-14. Epub 2015 Mar 29 PubMed.
- Oliveira FP, Castelo-Branco M. Computer-aided diagnosis of Parkinson's disease based on [(123)I]FP-CIT SPECT binding potential images, using the voxels-as-features approach and support vector machines. J Neural Eng. 2015 Apr;12(2):026008. Epub 2015 Feb 24 PubMed.
- Padilla P, Ramírez J, Górriz JM, Salas-González D, Alvarez-Illán I, Parkinson's Progression Markers Initiative. Statistical significance in the selection of the regions of interest for Parkinson brain image processing. Stud Health Technol Inform. 2014;207:19-26. PubMed.
- Kehler J, Nielsen J. PDE10A inhibitors: novel therapeutic drugs for schizophrenia. Curr Pharm Des. 2011;17(2):137-50. PubMed.
News
- Can Brain Metabolism Predict Huntington’s Symptoms?
- Study Finds Ways to Predict, Track Huntington’s Disease
- Deep Brain Stimulation: It’s All About the Rhythm
- FDA Gives a Nod for Alzheimer's and Parkinson's Biomarkers
- Tau PET Fits With CSF, Grows Over Time, Picks up Frontotemporal Cases
- References and Thresholds—Amyloid Imaging Protocols Debated at AAIC
- Can Brain Metabolism Predict Huntington’s Symptoms?
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.