Microglial Marker TREM2 Rises in Early Alzheimer’s and on Western Diet
Quick Links
Variants in the microglial receptor TREM2 boost the risk for Alzheimer’s disease, but the protein’s exact role remains something of a mystery. Two recent studies shed more light on the factors affecting TREM2. In the March 3 EMBO Molecular Medicine, researchers led by Christian Haass and Michael Ewers at Ludwig-Maximilians University, Munich, reported that levels of a soluble fragment of TREM2 in cerebrospinal fluid change over the course of Alzheimer’s disease, peaking at prodromal stages. Moreover, sTREM2 correlated with markers of neurodegeneration, hinting the two are connected. Meanwhile, researchers led by Gareth Howell at The Jackson Laboratory, Bar Harbor, Maine, uncovered a link between TREM2 and diet. They found that wild-type and AD mice eating chow that mimicked a typical Western diet not only developed neuroinflammation, but also had more TREM2-positive microglia in their brains than did mice on healthier feed. In addition, the AD mouse brains accumulated more amyloid plaque on Western fare. The research appeared in the February 18 Scientific Reports. Both studies strengthen the evidence for TREM2 activity in AD, but whether the protein protects or harms remains up in the air.
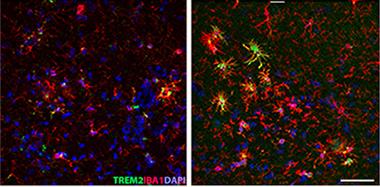
Diet-Related Inflammation.
More microglia expressing TREM2 (green) and Iba1 (red) crowd the hippocampi of APPPS1 mice eating a Western diet (right) than in those eating healthy chow (left). [Courtesy of Graham et al., Scientific Reports.]
Despite intense study in recent years, researchers still know little about TREM2’s role in AD. A few previous studies had examined CSF sTREM2 levels in AD patients, and their findings conflicted. Earlier work from Haass’ group reported lower CSF sTREM2 in AD patients than controls, while two other groups found it was higher (see Kleinberger et al., 2014; Jan 2016 news).
The new study from Haass and Ewer helps untangle the mystery by examining cross-sectional data from people at different stages of disease. First author Marc Suárez-Calvet compared CSF samples from 150 controls, 63 people with preclinical AD, 111 in prodromal stages, and 200 with dementia. Levels of sTREM2 were lowest in controls, slightly higher in the preclinical group, and highest in the prodromal population, but fell back toward control levels in those with dementia. In each group, sTREM2 correlated with elevated total tau and phospho-tau, which are considered markers of neurodegeneration. Strengthening this connection, the authors also saw higher levels of sTREM2 in 39 people with suspected non-AD pathology (SNAP). This group has high total tau and p-tau in the absence of amyloid markers. Notably, 103 people who had mild cognitive impairment without amyloid or tau pathology had normal TREM2 levels. Altogether, the results bolster the idea that TREM2 denotes neurodegeneration in general, but is not a specific marker for AD.
Carlos Cruchaga at Washington University in St. Louis, who led one of the other sTREM2 studies, found the new data suggesting dynamic changes in sTREM2 over the course of AD compelling. He said the findings help make sense of previous studies. “When you put everything together, the data are consistent. Depending on what population you are studying, you see higher or lower levels of sTREM2,” he told Alzforum. He noted that other CSF biomarkers, such as tau and the chemokine MCP1, demonstrate the same pattern of an early rise and late fall in AD, suggesting this may be a general phenomenon (see Mar 2014 news).
David Knopman at the Mayo Clinic in Rochester, Minnesota, however, noted that more data will be needed to parse out what TREM2 is doing. For one thing, it is unclear whether the rise in sTREM2 in prodromal AD exacerbates or results from pathology, he said. He was also struck by the fact that the authors found an increase in sTREM2 with age in all cohorts. “The study was well-done, but some of the results are puzzling. Because sTREM2 rises with age, it is not clear that the increase reflects neurodegeneration. Overall, I am intrigued by the behavior of sTREM2 in CSF and would like to see more work on it,” Knopman wrote to Alzforum.
The dietary study came at TREM2 from a different direction. Howell and colleagues were primarily interested in the effect that diet might have on aging and Alzheimer’s pathology. Several epidemiology studies have correlated diet with AD risk, and have reported protective effects in particular from a Mediterranean diet, which emphasizes fruit, vegetables, fish, and whole grains (see Apr 2006 news; Aug 2009 news; Valls-Pedret et al., 2015). On the other hand, mid-life obesity, which can result from a poor diet, raises AD risk.
To assess the effect of diet, the authors first formulated a mouse chow that mimicked the main features of standard Western fare. Previous diet studies had mostly investigated the effects of specific dietary components, such as high fat or high cholesterol, but such approaches do not reflect how most Westerners eat, Howell noted. He devised a chow that instead incorporated more animal-based fats and proteins along with refined sugar, while lacking fiber and some vitamins and minerals. By contrast, healthy mouse chow contains more plant-based proteins and fats and no added sugar. Total caloric intake of mice on the two diets were equivalent.
First author Leah Graham fed the Westernized chow to two-month-old wild-type B6 mice for eight months. The animals packed on the pounds, ending up 30 to 50 percent heavier than their healthy-eating peers. They also developed high blood sugar, though not diabetes. In their brains, the main effect was increased inflammation. The number of reactive astrocytes roughly doubled in the hippocampus and entorhinal cortex, while the number of activated and TREM2-positive microglia grew by around 20-30 percent. Although their brains remained largely normal in size and thickness overall, the mice did lose about 10 percent of their hippocampal neurons compared to controls.
The authors wondered what effect the Western diet would have on AD model mice. APPPS1 mice that gobbled the unhealthy chow for eight months also became obese and developed a similar pattern of neuroinflammation. Notably, the number of amyloid plaques in their hippocampi nearly doubled, and they sported about twice as many TREM2-positive microglia in their hippocampi as healthy-eating peers (see image above). They had greater numbers of activated microglia around plaques as well. Cell loss was similar to that in wild-type mice on the Western diet.
Frédéric Calon at Université Laval, Québec, found the work intriguing. He noted that the data agree with previous studies that reported a boost in activated astrocytes in animals on high-fat diets. “The reported increase in TREM2 in microglia/monocytes is particularly novel and compelling,” he wrote to Alzforum (see full comment below). He added that it would be interesting to look for potential changes in tau pathology in these mice as well.
Haass and Ewers saw additional possibilities in the data. “The results … suggest that TREM2 levels may be influenced by lifestyle factors such as diet, and thus offer the exciting perspective that TREM2 levels are modifiable and may become a treatment target in humans,” they wrote to Alzforum (see full comment below). However, they pointed out that researchers still do not know whether TREM2 helps or harms the brain. This basic question will have to be answered before the receptor could be targeted therapeutically.
For their part, Howell and colleagues would like to know whether the Western diet alone caused neuroinflammation, or whether obesity also plays a role. To dissect the contribution of each factor, they are feeding the Western diet to mouse strains that stay lean as well as those with a tendency to tubbiness, and examining what happens to their brains. They are also investigating the effect of exercise. In preliminary studies, physical activity appears to ameliorate the brain inflammation seen with poor diets. The scientists will also feed the unhealthy chow to mice that carry harmful TREM2 variants such as R47H, to better understand how the microglia protein interacts with pathology. “These studies may help us understand the interactions between genetic susceptibility and environmental risk factors such as diet and physical inactivity,” Howell told Alzforum.—Madolyn Bowman Rogers
References
News Citations
- TREM2 Goes Up in Spinal Fluid in Early Alzheimer’s
- DIAN Longitudinal Data Surprises With Late Drop in Tau
- Mediterranean Diet Slims Down Risk of AD
- Pass the Bordeaux—Mediterranean Diet, Exercise Reduce AD Risk
Research Models Citations
Paper Citations
- Kleinberger G, Yamanishi Y, Suárez-Calvet M, Czirr E, Lohmann E, Cuyvers E, Struyfs H, Pettkus N, Wenninger-Weinzierl A, Mazaheri F, Tahirovic S, Lleó A, Alcolea D, Fortea J, Willem M, Lammich S, Molinuevo JL, Sánchez-Valle R, Antonell A, Ramirez A, Heneka MT, Sleegers K, van der Zee J, Martin JJ, Engelborghs S, Demirtas-Tatlidede A, Zetterberg H, Van Broeckhoven C, Gurvit H, Wyss-Coray T, Hardy J, Colonna M, Haass C. TREM2 mutations implicated in neurodegeneration impair cell surface transport and phagocytosis. Sci Transl Med. 2014 Jul 2;6(243):243ra86. PubMed.
- Valls-Pedret C, Sala-Vila A, Serra-Mir M, Corella D, de la Torre R, Martínez-González MÁ, Martínez-Lapiscina EH, Fitó M, Pérez-Heras A, Salas-Salvadó J, Estruch R, Ros E. Mediterranean Diet and Age-Related Cognitive Decline: A Randomized Clinical Trial. JAMA Intern Med. 2015 Jul;175(7):1094-103. PubMed.
External Citations
Further Reading
News
- Enter the New Alzheimer’s Gene: TREM2 Variant Triples Risk
- Fall Flurry of Letters Kicks Up Dust Around TREM2
- TREM2 Variant Doubles the Risk of ALS
- TREM2 Mystery: Altered Microglia, No Effect on Plaques
- TREM2 Data Surprise at SfN Annual Meeting
- United in Confusion: TREM2 Puzzles Researchers in Taos
- TREM2 Buoys Microglial Disaster Relief Efforts in AD and Stroke
- TREM2 Tidbits at AAIC: Genetics, Clinical Data
Primary Papers
- Suárez-Calvet M, Kleinberger G, Araque Caballero MÁ, Brendel M, Rominger A, Alcolea D, Fortea J, Lleó A, Blesa R, Gispert JD, Sánchez-Valle R, Antonell A, Rami L, Molinuevo JL, Brosseron F, Traschütz A, Heneka MT, Struyfs H, Engelborghs S, Sleegers K, Van Broeckhoven C, Zetterberg H, Nellgård B, Blennow K, Crispin A, Ewers M, Haass C. sTREM2 cerebrospinal fluid levels are a potential biomarker for microglia activity in early-stage Alzheimer's disease and associate with neuronal injury markers. EMBO Mol Med. 2016 May 2;8(5):466-76. PubMed.
- Graham LC, Harder JM, Soto I, de Vries WN, John SW, Howell GR. Chronic consumption of a western diet induces robust glial activation in aging mice and in a mouse model of Alzheimer's disease. Sci Rep. 2016 Feb 18;6:21568. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Laval University Research Center
The study is interesting, as it further confirms the importance of diets on markers related to AD neuropathology. It is focused on glial activation, which makes sense, as it replicates previous work showing high energy intake from fat increases GFAP (Buckman et al., 2015; Valdearcos et al., 2014; Julien et al., 2010).
The reported increase in TREM2 in microglia/monocytes is particularly novel and compelling, given recent publications in human CSF and animal models (Jay et al., 2015; Wang et al., 2015; St-Amour et al., 2015; Piccio et al., 2016).
Concerning the diet, I am not sure that a "true Western diet" is that well-defined, particularly when treating a mouse. Unfortunately, perhaps due to a lack of space, the authors do not describe the diet they have used very well (no detailed table of contents, at least in the PDF version I have). Thus it is hard to compare with previous work and to identify key dietary variables.
It would have been nice to look at tau pathology as well and to confirm peripheral metabolic impairments (glucose intolerance, insulin resistance, etc.) in the same animals.
References:
Buckman LB, Thompson MM, Lippert RN, Blackwell TS, Yull FE, Ellacott KL. Evidence for a novel functional role of astrocytes in the acute homeostatic response to high-fat diet intake in mice. Mol Metab. 2015 Jan;4(1):58-63. Epub 2014 Oct 16 PubMed.
Valdearcos M, Robblee MM, Benjamin DI, Nomura DK, Xu AW, Koliwad SK. Microglia dictate the impact of saturated fat consumption on hypothalamic inflammation and neuronal function. Cell Rep. 2014 Dec 24;9(6):2124-38. Epub 2014 Dec 11 PubMed.
Julien C, Tremblay C, Phivilay A, Berthiaume L, Emond V, Julien P, Calon F. High-fat diet aggravates amyloid-beta and tau pathologies in the 3xTg-AD mouse model. Neurobiol Aging. 2010 Sep;31(9):1516-31. Epub 2008 Oct 15 PubMed.
Jay TR, Miller CM, Cheng PJ, Graham LC, Bemiller S, Broihier ML, Xu G, Margevicius D, Karlo JC, Sousa GL, Cotleur AC, Butovsky O, Bekris L, Staugaitis SM, Leverenz JB, Pimplikar SW, Landreth GE, Howell GR, Ransohoff RM, Lamb BT. TREM2 deficiency eliminates TREM2+ inflammatory macrophages and ameliorates pathology in Alzheimer's disease mouse models. J Exp Med. 2015 Mar 9;212(3):287-95. Epub 2015 Mar 2 PubMed.
Wang Y, Cella M, Mallinson K, Ulrich JD, Young KL, Robinette ML, Gilfillan S, Krishnan GM, Sudhakar S, Zinselmeyer BH, Holtzman DM, Cirrito JR, Colonna M. TREM2 lipid sensing sustains the microglial response in an Alzheimer's disease model. Cell. 2015 Mar 12;160(6):1061-71. Epub 2015 Feb 26 PubMed.
St-Amour I, Cicchetti F, Calon F. Immunotherapies in Alzheimer's disease: Too much, too little, too late or off-target?. Acta Neuropathol. 2016 Apr;131(4):481-504. Epub 2015 Dec 21 PubMed.
Piccio L, Deming Y, Del-Águila JL, Ghezzi L, Holtzman DM, Fagan AM, Fenoglio C, Galimberti D, Borroni B, Cruchaga C. Cerebrospinal fluid soluble TREM2 is higher in Alzheimer disease and associated with mutation status. Acta Neuropathol. 2016 Jun;131(6):925-33. Epub 2016 Jan 11 PubMed.
Make a Comment
To make a comment you must login or register.