Plaque Microglia—Mobile But Limited
Quick Links
Microglia are rather cozy with amyloid plaques, but how they snuggle up to these protein deposits and what ensues thereafter is not well understood. In the April 16 Journal of Neuroscience, researchers led by Michael Calhoun at the University of Tuebingen, Germany, give a vivid account of the microglia/plaque relationship. Their time-delay images show that dynamic microglia play an active role in seeking out plaques and engulfing Aβ. The cells’ prowess is not without limitation, however. “One surprising thing we found was there is really a recruitment issue, in that regardless of plaque size the number of microglia that surround individual plaques reach an upper limit,” said Calhoun in an interview with ARF. Understanding and overcoming that limitation is one idea worth thinking about in the context of therapy, he added.
The glia, in fact, may already be overcoming that limitation themselves—by beefing up. First author Tristan Bolmont and colleagues found that individual microglia can increase in size when they contact plaques, and that larger plaques consorted with larger microglia. The findings are based on multiphoton microscopy of mouse brain taken at intervals through a cranial window. The researchers used three different fluorescent markers to visualize microglia (expressing green fluorescent protein driven by the Iba-1 microglial promoter), amyloid (labeled with methoxy –X04, a Congo red derivative), and blood vessels (labeled with Texas red dextran). By using software to keep track of major landmarks in the field of view, including the blood vessels, the researchers were able to come back time and again to the same plaques to record microglial activity (see figure below).
With the ability to re-image the same location in the brain, the researchers have been able to begin to quantify the glia/plaque relationship. They found that the volume of small- and medium-sized plaques increases over a month in three-month-old transgenic animals (APPPS+ mice), while the volume of large plaques actually decreases. The number of microglia surrounding the plaques increases over that same time interval, but as Calhoun noted, there is a limit to the density of the glia surrounding plaques.
The researchers found that the microglia are incredibly dynamic, morphing in both position and size (see ARF related news story). In APPPS+ mice, for example, about half of the microglia that lie close to plaques migrate so that their cell bodies are in immediate contact with the deposits. This migration typically took one to two days, as seen in the figure below.
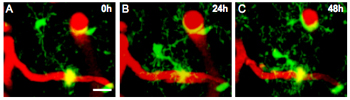
Microglia on the Move
A microglia (green) making contact with an amyloid plaque (yellow) via long cellular processes moves in to cover the protein deposit over the course of two days. Blood vessels are seen as red fluorescence. Image credit: Michael Calhoun
The data indicate that resting microglia can respond to their environment. The glia seem to first become polarized, extending processes in the direction of the plaque, then, on finally reaching it, they become enlarged. That process plays out in the movie below.
Polarized processes aid movement of resident microglia around amyloid
This movie shows the dynamic nature of microglia, which extend and retract processes to the amyloid plaque (center) as if sampling the plaque environment. Movie credit: Michael Calhoun
The findings help address one of the important and oft-debated questions about plaque microglia, namely, where do they come from? There are indications that some of them might come from the blood (see ARF related news story), and some through division of existing cells. “We were able to show that some of them actually transform from normal resting microglia into a more macrophagic type of microglia that surrounds plaque,” said Calhoun. He added that figuring out percentages of where all the cells come from needs more quantitative study. “But I think it is interesting to know that you can transform these resting microglia,” he said.
What does this all mean for plaque removal? The findings show a dynamic response to plaques. Microglia both actively migrated to plaques—presumably to help control plaque growth since the researchers found that the glia actively engulfed Aβ—and grew in size. Microglia surrounding medium-sized plaques were some 35 percent bigger than those associated with small plaques, for example, while microglia on large plaques were more than twice the size of those on medium plaques. Interestingly, the number of adjacent glia is also indicative of whether plaques will increase or decrease in size; more glia means either a smaller increase or a decrease. “This is both predictive and correlative, so from microglia coverage we can determine the likelihood of what the plaque will do,” said Calhoun.
However, despite the microglia’s rallying around plaques, the researchers never saw complete plaque removal. That is consistent with the observation that when Aβ production is halted in transgenic mice, plaques do not disappear (see ARF related news story). “That makes me wonder if there is homeostasis, or if the microglia at some point need an additional stimulus to get at the plaques,” said Calhoun. Brad Hyman, Massachusetts General Hospital, Boston, agrees. “I think this [study] is really intriguing and agrees quite well with our hypothesis that microglia respond to plaques but are not able to ingest them under ordinary circumstances,” he e-mailed ARF. Hyman recently used similar imaging techniques to show that amyloid plaques pop up quite rapidly in the brain (see ARF related news story), a phenomenon that Bolmont and colleagues also recorded.
Calhoun said it is not clear how one might coax microglia to completely remove plaques and take their contents away into the blood, but he thinks that some microglia may be capable or that some factors might persuade them. “We need to look at the heterogeneity in microglia and what molecular factors we can use to drive that process,” he said.—Tom Fagan
References
News Citations
Further Reading
Primary Papers
- Bolmont T, Haiss F, Eicke D, Radde R, Mathis CA, Klunk WE, Kohsaka S, Jucker M, Calhoun ME. Dynamics of the microglial/amyloid interaction indicate a role in plaque maintenance. J Neurosci. 2008 Apr 16;28(16):4283-92. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Harvard Medical School
This paper illustrates nicely how complex and dynamic the interactions between microglia and β amyloid are, and how relevant these interactions are to the pathogenesis of Alzheimer disease, at least in mouse models. The paper supports a role for microglia in clearing β amyloid and suggests that acute as well as chronic/long-term interactions of these cells with plaques are important and need to be investigated. I believe two important questions raised again by the paper (as well as by other papers) need to be addressed in future studies:
1. How do microglia accumulate in and around plaques? There is compelling data in the literature that support a migratory event involving recruitment to plaques, but perhaps also there is a small element of proliferation, though the question of proliferation needs to be further explored to be proven.
2. What are the molecular mechanisms by which microglia promote clearance of β amyloid and why do we still get plaques in spite of this microglial role?
University of Southern California
The paper by Bolmont and colleagues (2008) represents an elegant set of experiments designed to track microglia in a doubly-transgenic mouse model of AD. The authors crossed an Iba1-GFP transgenic (thereby labeling microglia green) with a co-injected APP/PS1 AD mouse and imaged cerebral vessels, microglia, and amyloid plaques using multi-photon microscopy, by way of a cranial window. They imaged these animals at short-time intervals (within minutes), and over longer time periods (from days to one month). In my view, there are a number of important take-home messages, and also a whole host of interesting questions raised by this work.
Importantly, the authors found in their “longer time period” imaging experiments that amyloid plaques are remarkably stable, as noted also by Joanna Jankowsky and David Borchelt using a tet-inducible AD mouse model (Jankowsky et al., 2005). This is an interesting result in and of itself, because an earlier view was that cerebral amyloid deposits were dynamic, coming and going based on changes in microenvironment. The authors have gone further by showing that microglia migrate toward amyloid deposits, and once they reach their destination, they remain there as permanent residents and enlarge their somas. While the authors do not report fixed tissue-immunostaining of this cohort of microglia with activation makers (e.g., CD11b/Mac-1, CD45, F4/80 Ag, etc.), by inference it is highly likely that these cells represent the “activated” microglia typically found in close vicinity to mature amyloid plaques, which are undergoing an anti-phagocytic, proinflammatory innate immune response (Town et al., 2005). This is further supported by the authors’ finding of reduced fine processes for microglia “on” plaques (probably representative of the activated, “amoeboid” morphology seen by others). The authors also found that microglia volume increased in proportion with amyloid plaque volume, providing strong evidence, in my view, that larger, “mature” amyloid plaques are more immunogenic than less mature plaques. This was most obvious when considering “medium-sized” versus “large” amyloid plaques, where the latter showed a 225 percent increase in microglia volume.
I find most intriguing the authors’ finding that microglia surrounding amyloid plaques displayed puncta of the dye used for Aβ imaging, which generally persisted throughout the duration of the experiments. Pioneering early reports from Henry Wisniewski and Jerzy Wegiel demonstrated at the ultrastructural level that microglia around plaques fail to internalize amyloid fibrils (Wisniewski et al., 1989; Wegiel and Wisniewski, 1990; Wisniewski and Wegiel, 1994). More recent reports (Stalder et al., 1999; 2001) have further highlighted that fibrillar amyloid is not present within microglia, seemingly at odds with the authors’ finding. However, as the authors rightly point out, differences in methodology (not the least of which is in-vivo imaging versus postmortem analysis of tissue sections) may account for this. Assuming this result is robust, it is interesting that these puncta generally remained visible throughout the course of the experiment, suggesting that even if microglia are capable of internalizing amyloid deposits in vivo, they are not efficient amyloid degraders. This begs the critically important question of how to promote efficient microglia-mediated clearance of amyloid plaques as a potential therapeutic modality, which is something that we are also intensely interested in (Town et al., 2005).
A number of interesting questions arise from this paper. Throughout the course of their long-term imaging experiments, did the authors detect new, rapidly forming plaques as reported recently by Brad Hyman’s group (Meyer-Luehmann et al., 2008)? If so, did microglia migrate to these plaques with differing kinetics from already-formed ones? Also, the authors mention the important point that they cannot discriminate between newly emigrating blood-derived monocytes/macrophages and resident microglial cells; however, they do report that, while small plaques increased in size by 84 percent, large plaques actually decreased in size by 12 percent. If not due to measurement error, one possibility is that the reduced size of large plaques could be due to infiltrating monocytes/macrophages, which may be more tuned to remove amyloid plaques.
References:
Jankowsky JL, Slunt HH, Gonzales V, Savonenko AV, Wen JC, Jenkins NA, Copeland NG, Younkin LH, Lester HA, Younkin SG, Borchelt DR. Persistent amyloidosis following suppression of Abeta production in a transgenic model of Alzheimer disease. PLoS Med. 2005 Dec;2(12):e355. Epub 2005 Nov 15 PubMed.
Meyer-Luehmann M, Spires-Jones TL, Prada C, Garcia-Alloza M, de Calignon A, Rozkalne A, Koenigsknecht-Talboo J, Holtzman DM, Bacskai BJ, Hyman BT. Rapid appearance and local toxicity of amyloid-beta plaques in a mouse model of Alzheimer's disease. Nature. 2008 Feb 7;451(7179):720-4. PubMed.
Stalder M, Phinney A, Probst A, Sommer B, Staufenbiel M, Jucker M. Association of microglia with amyloid plaques in brains of APP23 transgenic mice. Am J Pathol. 1999 Jun;154(6):1673-84. PubMed.
Stalder M, Deller T, Staufenbiel M, Jucker M. 3D-Reconstruction of microglia and amyloid in APP23 transgenic mice: no evidence of intracellular amyloid. Neurobiol Aging. 2001 May-Jun;22(3):427-34. PubMed.
Town T, Nikolic V, Tan J. The microglial "activation" continuum: from innate to adaptive responses. J Neuroinflammation. 2005 Oct 31;2:24. PubMed.
Wisniewski HM, Wegiel J, Wang KC, Kujawa M, Lach B. Ultrastructural studies of the cells forming amyloid fibers in classical plaques. Can J Neurol Sci. 1989 Nov;16(4 Suppl):535-42. PubMed.
Wegiel J, Wisniewski HM. The complex of microglial cells and amyloid star in three-dimensional reconstruction. Acta Neuropathol. 1990;81(2):116-24. PubMed.
Wisniewski HM, Wegiel J. The role of microglia in amyloid fibril formation. Neuropathol Appl Neurobiol. 1994 Apr;20(2):192-4. PubMed.
View all comments by Terrence TownUniversity of Antwerp
This is a fascinating paper from the group of Mathias Jucker and Michael Calhoun studying in vivo interaction of amyloid plaques and their most intriguing and prominent cellular component—the microglia. Employing two-photon imaging of GFP-labeled microglia, methoxy-X04-labeled plaques, and triangulating on the study areas for repeated measures with the help of dextran conjugate-labeled blood vessels, the authors support and extend previous observations (Davalos et al., 2005; Nimmerjahn et al., 2005; Meyer-Luehmann et al., 2008) of a highly dynamic role of microglia in conducting brain surveillance. Like a true vigilante, supposedly “resting” microglia continually patrol their microenvironment with extremely motile ramified cellular processes. At the first contact of amyloid plaques, they jump on-scene and try to clear the plaques and, expectedly, do some collateral damage to the surrounding environment.
One of the many reasons I find this study fascinating is that it tries to “assess” not only how many microglia arrive at the sites of plaques, but also how fast they do so, and how successfully they limit or resolve these plaques. Though not quantitatively, the study also tries to find in vivo evidence of Aβ phagocytosis by microglia.
For instance, on the high-resolution time-lapse images acquired over minutes to hours or days, about half of the studied microglia were shown to migrate to the proximity of the amyloid plaques within 24-48 hours. These data seem to differ from some other studies where only microglial processes were observed to be dynamic but not their somas (Davalos et al., 2005; Nimmerjahn et al., 2005). However, these prior studies are based on brain injury models, and the impact of such injuries might be different from less acute but difficult-to-heal Aβ plaques.
Movement of microglial cell bodies to the site of amyloid plaques has also been observed to occur within 24 hours in a recently published study (Meyer-Luehmann et al., 2008). It is possible that a tighter and more “stable” cell soma-Aβ contact, as shown in the current paper, is necessary for attempted walling off and/or phagocytosis by microglia. In contrast to Meyer-Luehmann et al. (2008), but similar to several previous histology-based studies, the authors also showed globular intracellular methoxy-X04 labeling within microglia, which on parallel histology studies colocalized with the lysosomal marker lamp-1. However, whether these mononuclear phagocytes are bone marrow (BM)-derived or resident microglia remains an open question. BM-derived macrophages have been recently shown to infiltrate rodent brain in response to injected Aβ40 and Aβ42, and they were further able to phagocytose amyloid, while resident microglia did not appear to do (Simard et al., 2006).
How about the age-old question, Is microglia-Aβ interaction harmful or beneficial? The current study and similar studies showing a role of microglia in walling off the injured areas and in limiting plaque growth clearly point towards a beneficial effect (Simard et al., 2006; Bolmont et al., 2008). Deficiency of Ccr2, a chemokine receptor expressed on microglia, accelerating early disease progression in rodents, also supports these data (El Khoury et al., 2007). But we have to keep in mind that the rodent data have still to be correctly translated to humans. For instance, controlled human clinical trials have repeatedly shown less robust effects of anti-inflammatory drugs in contrast to their fantastic beneficial effects on mouse models. Some of the issues raised in the current paper, such as dynamics of plaque growth due to soluble Aβ, is already one factor that would be different in humans. Thus, while not all is answered, the in vivo approach employed in some of these elegant papers is definitely progress in the right direction.
References:
Davalos D, Grutzendler J, Yang G, Kim JV, Zuo Y, Jung S, Littman DR, Dustin ML, Gan WB. ATP mediates rapid microglial response to local brain injury in vivo. Nat Neurosci. 2005 Jun;8(6):752-8. PubMed.
El Khoury J, Toft M, Hickman SE, Means TK, Terada K, Geula C, Luster AD. Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nat Med. 2007 Apr;13(4):432-8. PubMed.
Meyer-Luehmann M, Spires-Jones TL, Prada C, Garcia-Alloza M, de Calignon A, Rozkalne A, Koenigsknecht-Talboo J, Holtzman DM, Bacskai BJ, Hyman BT. Rapid appearance and local toxicity of amyloid-beta plaques in a mouse model of Alzheimer's disease. Nature. 2008 Feb 7;451(7179):720-4. PubMed.
Nimmerjahn A, Kirchhoff F, Helmchen F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science. 2005 May 27;308(5726):1314-8. PubMed.
Simard AR, Soulet D, Gowing G, Julien JP, Rivest S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer's disease. Neuron. 2006 Feb 16;49(4):489-502. PubMed.
View all comments by Samir Kumar-SinghUniversity of California San Diego
This paper by the group of Michael Calhoun confirms, in vivo, a role for microglia in amyloid plaque maintenance. The authors succeeded in monitoring live changes in plaque size, and correlate them to changes in number and appearance of “on site” microglial cells. Interestingly, they describe microglial uptake of the amyloid-β fibrils bound to the methoxy-X04 dye. This is an unexpected result that contradicts previous reports (Stalder et al., 2001). However, if indeed microglial cells can phagocytose Aβ fibrils, why are these cells not able to completely disassemble or clear Aβ plaques? Instead, microglial cells seem to just guarantee a stable plaque size after their initial acute growth, as previously suggested by Meyer-Luehmann et al. (2008). Animal species differences should be also taken into account, since findings for human microglia cells can contrast with the one for murine microglial cells (Blasi et al., 1995).
Overall, this paper opens the door to understanding the mechanism with which microglia would control the size of amyloid-β plaques and therefore to new possible therapeutic approaches.
References:
Blasi E, Barluzzi R, Mazzolla R, Tancini B, Saleppico S, Puliti M, Pitzurra L, Bistoni F. Role of nitric oxide and melanogenesis in the accomplishment of anticryptococcal activity by the BV-2 microglial cell line. J Neuroimmunol. 1995 Apr;58(1):111-6. PubMed.
Meyer-Luehmann M, Spires-Jones TL, Prada C, Garcia-Alloza M, de Calignon A, Rozkalne A, Koenigsknecht-Talboo J, Holtzman DM, Bacskai BJ, Hyman BT. Rapid appearance and local toxicity of amyloid-beta plaques in a mouse model of Alzheimer's disease. Nature. 2008 Feb 7;451(7179):720-4. PubMed.
Stalder M, Deller T, Staufenbiel M, Jucker M. 3D-Reconstruction of microglia and amyloid in APP23 transgenic mice: no evidence of intracellular amyloid. Neurobiol Aging. 2001 May-Jun;22(3):427-34. PubMed.
Retired psychiatrist and Alzheimers Disease student
This research design could provide a means of exploring curcumin's potential role as a facilitator of microglial phagocytosis and degradation of amyloid plaque.
View all comments by Stephen StatenMake a Comment
To make a comment you must login or register.