Umbilical Cord With Presenilin Mutation Births New Cell Model of Familial AD
Quick Links
Induced pluripotent stem cell models of Alzheimer’s disease have become important because they allow researchers to study human neurons carrying AD mutations. However, iPSC lines take weeks to establish, require viral vectors to reprogram adult cells—and the required skin sample leaves behind a punch hole that’s not every tissue donor’s cup of tea. In the May 21 PLOS ONE, Marlene Jimenez-Del-Rio at the University of Antioquia in Medellin, Colombia, describes an alternative method.
- Stem cells from umbilical cord quickly differentiated into neuron-like cells.
- Those with the E280A presenilin 1 mutation produced more Aβ42.
- The cells also accumulated more phosphorylated tau and markers of apoptosis.
The researchers took mesenchymal stromal stem cells from the umbilical cord of a baby carrying the Paisa E280A mutation in presenilin 1. After a week in culture with a mix of growth factors, the cells expressed cholinergic markers. Lo and behold, these E280A neurons produced more Aβ42 than did a control line. They also accumulated phosphorylated tau, reactive oxygen species, and apoptotic markers.
“Using a natural biological material that was not genetically manipulated, we can recreate the neuropathological features of a multifactorial disease such as familial AD in as short a time as 11 days,” Jimenez-Del-Rio wrote to Alzforum. She believes these cells will be valuable for mechanistic studies.
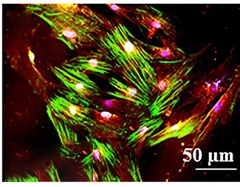
A Model in a Week? Umbilical cord stem cells subjected to a differentiation cocktail for a week express neuronal markers MAP2 (green) and choline acetyl transferase (red). [Courtesy of Soto-Mercado et al., PLoS One.]
Others agreed that the method has potential. “The cells are a useful reagent,” Ashley Bush at the University of Melbourne, Australia, wrote to Alzforum. Russell Swerdlow at University of Kansas Medical Center in Kansas City said the study extends previous findings on the presenilin 1 E280A mutation. “The authors do a nice job of demonstrating the mutation leads to cell stress and recapitulates molecular phenomena associated with AD,” he wrote.
The Paisa mutation is named for the large Colombian kindred, about 5,000 strong, in whom this genetic variant is prevalent (Mar 2010 news). The aberrant enzyme overproduces Aβ42, and the brains of carriers develop slightly differently from those of noncarriers, with mutation effects discernable at ages as young as 9 (Jul 2015 news). The kindred has made invaluable contributions to research, participating in a secondary prevention trial as well as observational and genetic studies (Aug 2019 news; Aug 2019 news; Nov 2019 news).
In Jimenez-Del-Rio’s study, pregnant women in this kindred agreed to donate umbilical cords after birth. The researchers obtained cords from the births of 10 healthy babies in this population. One of them carried the E280A mutation. First author Viviana Soto-Mercado harvested a 3-inch length of umbilical cord from this carrier and the same from the cord of a noncarrier. She dissociated the cells and cultured them in medium containing fibroblast growth factor, retinoic acid, and sonic hedgehog peptide, a cocktail that promotes neuronal differentiation. After a week, the cells expressed the neuronal markers MAP2 and β tubulin III, as well as the cholinergic neuron markers choline-acetyltransferase and acetylcholinesterase (see image above).
Two days later, the E280A cholinergic-like neurons contained deposits that bound the antibody 6E10. It detects Aβ, amyloid precursor protein (APP), and products of α- and β-secretase. Mass spectrometry revealed these intracellular inclusions mostly consisted of the β-secretase C-terminal fragment (β-CTF). No such deposits appeared in wild-type cholinergic-like neurons, nor in the parent wild-type or E280A mesenchymal stromal stem cells.
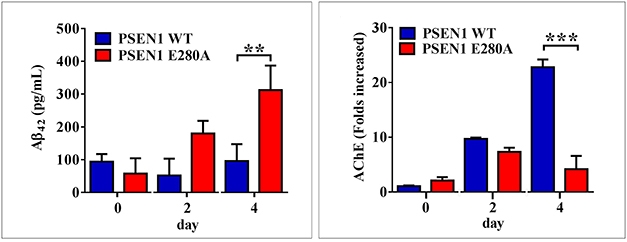
Fast Difference. Four days after differentiation, neurons carrying E280A (red) make more Aβ42 (left) and less acetylcholinesterase (right) compared with wild-types (blue). [Courtesy of Soto-Mercado et al., PLOS ONE.]
By day 11 in culture, E280A cholinergic-like neurons secreted three times as much extracellular Aβ42 as did wild-types (see image above). It is unclear why β-CTF accumulated before Aβ42, but E280A is known to reduce Aβ production while raising the Aβ42/40 ratio (Li et al., 2016; Sun et al., 2017). Jimenez-Del-Rio speculated that the E280A presenilin 1 might alter the cellular location of γ-secretase in such a way as to promote first buildup of intracellular APPβ and then Aβ42 production.
Starting at day 11, the cells developed the second hallmark of AD, i.e. tau phosphorylated at Ser202 and Thr205. Inhibiting the tau kinase JNK prevented this.
Other differences between mutant and wild-type quickly emerged as well. The E280A cholinergic-like neurons had more reactive oxygen species than wild-types on day seven, and lower mitochondrial membrane potential by day nine. The mutant cells accumulated several markers of apoptosis such as c-JUN, p53, and caspase 3 over their first 11 days in culture. They overreacted to acetylcholine, releasing more intracellular calcium than wild-types, and their acetylcholinesterase activity was weak.
Altogether, the data suggest that the buildup of intracellular Aβ products kicks off oxidative stress and toxicity, leading to the eventual death of cholinergic neurons, Jimenez-Del-Rio said. β-CTF has been associated with cellular toxicity before (Aug 2019 news). In future work, Jimenez-Del-Rio will test potential therapeutic interventions in this model.
She also plans to correct the E280A mutation with CRISPR so she can compare corrected and uncorrected cells. The E280A cell line she used carries an ApoE4 mutation, while the control line does not. However, Jimenez-Del-Rio believes this would not influence the cholinergic-like neurons much, since ApoE is expressed primarily by astrocytes. Bush suggested inhibiting γ-secretase in these cells to confirm that the observed changes are a direct result of increased Aβ42 production.
Gunnar Gouras at Lund University, Sweden, noted that the cholinergic-like neurons in these studies retain an immature phenotype, lacking the extensive dendritic trees of mature cells. He suggested it would be interesting to culture them for longer time periods to see if they develop mature processes and synapses.
Gouras agrees that umbilical cord mesenchymal stromal stem cells could have advantages over iPSC technology, including their ability to quickly differentiate into multiple cell types. In addition to cholinergic-like neurons, the authors produced adipocytes, osteoblasts, and chondrocytes from these stem cells. “I see this as another potential tool,” Gouras said.—Madolyn Bowman Rogers
References
Mutations Citations
News Citations
- Phoenix: Trials in Colombia and the U.S. for Those at Highest Risk?
- Familial Alzheimer’s Gene Alters Children’s Brains
- Crenezumab Update: Baseline Data from Colombian Prevention Trial
- Colombian Cohort Delivers Data on Blood NfL
- Can an ApoE Mutation Halt Alzheimer’s Disease?
- Familial AD Mutations, β-CTF, Spell Trouble for Endosomes
Antibody Citations
Paper Citations
- Li N, Liu K, Qiu Y, Ren Z, Dai R, Deng Y, Qing H. Effect of Presenilin Mutations on APP Cleavage; Insights into the Pathogenesis of FAD. Front Aging Neurosci. 2016;8:51. Epub 2016 Mar 11 PubMed.
- Sun L, Zhou R, Yang G, Shi Y. Analysis of 138 pathogenic mutations in presenilin-1 on the in vitro production of Aβ42 and Aβ40 peptides by γ-secretase. Proc Natl Acad Sci U S A. 2017 Jan 24;114(4):E476-E485. Epub 2016 Dec 5 PubMed.
Further Reading
Primary Papers
- Soto-Mercado V, Mendivil-Perez M, Velez-Pardo C, Lopera F, Jimenez-Del-Rio M. Cholinergic-like neurons carrying PSEN1 E280A mutation from familial Alzheimer's disease reveal intraneuronal sAPPβ fragments accumulation, hyperphosphorylation of TAU, oxidative stress, apoptosis and Ca2+ dysregulation: Therapeutic implications. PLoS One. 2020;15(5):e0221669. Epub 2020 May 21 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Kansas
This study extends prior cell culture, sub-cellular biochemical, animal, and human studies that show presenilin 1 mutations have biological consequences. As demonstrated nicely here, the stem-cell-derived model can facilitate studies of how specific PS1 mutations affect cells. The authors do a nice job of demonstrating the mutation leads to cell stress, and recapitulates molecular phenomena associated with AD.
It was certainly interesting to see the authors speculate on potential mechanisms. In this case, the scenario laid out in the discussion sought to reconcile the observed mutation-dependent cell phenomena with the amyloid cascade hypothesis. This led to speculation that the presenilin mutation increased intracellular amyloid-β oligomers, which subsequently acted as the agent of cell stress.
A rigorous assessment of whether Aβ oligomers are a critical mediator of the various cell changes observed, or are an additional byproduct of cell stress, a link in a longer mechanistic chain, or simply an independent byproduct of the mutant presenilin would be interesting to experimentally explore.
Make a Comment
To make a comment you must login or register.