Voilà SorLA! Sorting Receptor’s Structure Solved
Quick Links
Researchers have published the crystal structure of part of the SorLA neuronal sorting receptor, a known genetic risk factor for Alzheimer’s disease. Reporting in the February 2 Nature Structural and Molecular Biology, scientists led by Junichi Takagi of Osaka University, Suita, Japan, described the protein’s extracellular Vps10p domain. This region appears to capture Aβ by β-sheet extension, meaning it attracts peptides that form β sheets with similar sheets of its own. The structure lends support to the group’s recent hypothesis that SorLA binds Aβ monomers and escorts them to the lysosome for degradation, the authors write.
“The study of SorLA’s structure may help to develop small molecules to boost its function,” suggested Ekaterina Rogaeva of the University of Toronto.
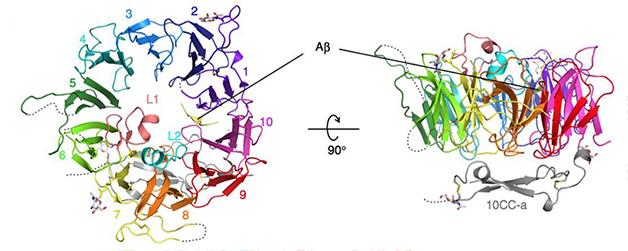
SorLA’s Aβ trap: The 10 "blades" of Vps10p form a tunnel. When Aβ (yellow) binds inside, loops L1 and L2 block the tunnel entrance (top view, left), while the 10CC region extends down like a lever (side view, right). This frees a proline residue that helps hold Aβ in place. [Image courtesy of Kitago et al., Nature Structural & Molecular Biology]
SorLA is short for sortilin-related receptor with low-density lipoprotein receptor class A repeats. The protein also goes by SORL1 and LR11. Previous studies reported that it is a genetic risk factor for both late-onset and early onset Alzheimer’s disease (see Alzgene entry; Jan 2007 news; Apr 2012 news). SorLA shuttles the amyloid precursor protein (APP) around the cell and reduces its processing by secretases (see Mar 2005 conference news). Takagi, along with Thomas Willnow of the Max-Delbrück-Center for Molecular Medicine, Berlin, and colleagues recently reported that SorLA’s Vps10p domain binds Aβ monomers as they are created and directs them to lysosomes for degradation (see Feb 2014 news). A familial AD mutation in this domain, G511R, impairs that clearance. In the current paper, joint first authors Yu Kitago, Masamichi Nagae, and Zenzaburo Nakata wanted to understand how Vps10p bound Aβ. They determined the crystal structures of Vps10p alone and when it was bound to the peptide.
Without a ligand, the Vps10p domain assumes a shape like a ship's propeller with 10 blades (see image above). Each blade forms a small β-sheet, a structure in which several polypeptide strands are held together by hydrogen bonds. Together they create a cone-shaped tunnel. An elongated region, called 10CC, wraps around the narrow end of the cone. Crystals of the empty protein formed only at low pH, suggesting this conformation occurs in acidic places like the lysosome. That supports the idea that the Vps10p domain releases its ligands there.
On the other hand, a fragment of Aβ, Aβ6-15, bound only at more neutral pH, above 5.5, implying that it attaches outside the lysosome. The peptide latched onto the edge of the first blade of the propeller, the most hydrophobic area of the tunnel’s surface, via hydrogen bonds. With the peptide bound, two loops on Vps10p, L1 and L2, formed a lid over part of the tunnel and pushed the peptide against the tunnel wall, seemingly holding it in place. If the researchers removed either loop, the Aβ6-15 failed to bind. The authors noted that the G511R mutation occurs adjacent to the L2 loop, which could disturb Aβ-binding. Vps10p’s 10CC region also jutted out from underneath the ring, like a lever. Without its electrostatic pull on the Vps10p tunnel, proline 590 was free to help hold Aβ in place.
The researchers then obtained the crystal structure for Vps10p when it bound to another of its ligands, a piece cleaved from SorLA’s propeptide. This fragment binds to Vps10p when the protein first matures to prevent premature binding of other peptides. The propeptide ligand occupied the same spot on blade 1 and caused SorLA to adopt the same conformation. Both this propeptide and Aβ form β-sheets on their own, and each sidled up to the β-sheet structure of the first blade, regardless of the specific sequence of amino acids. This suggests that SorLA recognizes and binds a variety of β-sheet forming peptides by β-sheet extension.
Takagi hypothesized that Aβ40, Aβ42, modified forms like pGluAβ, and other species of Aβ that share the Aβ6-15 fragment bind to Vps10p the same way. He and his co-authors are testing whether binding affinities differ among any of these forms of the Aβ peptide.
“Identifying the structure of any protein helps clarify its function,” wrote Scott Small, Columbia University, New York, in an email. He added that since mutations in SorLA linked to late-onset AD appear to affect APP processing, and those linked to early onset seem to reduce Aβ clearance, this difference may hint at distinct cell biology for each form of disease. Rogaeva noted that a number of other mutations, including those outside the Vps10p domain, have been associated with Alzheimer’s (see Vardarajan et al., 2014). It is possible that those mutations act by different mechanisms than G511R, she said.
Olav Andersen, Aarhus University, Denmark, remains unconvinced that SorLA serves to transport newly generated Aβ to the lysosome. Nevertheless, “the structure of the VPS10p domain is beautiful and the novelty in describing a flexible 10CC region is extremely interesting,” he remarked in an email.—Gwyneth Dickey Zakaib
References
Alzpedia Citations
News Citations
- SORLA Soars—Large Study Links Gene to Late-onset AD
- New Genetic Insights Into AD: SORL1 and Natural Selection
- Sorrento: Sorting Out Shedding of Ectodomains
- SORLA Serves Up Aβ for Destruction
Paper Citations
- Vardarajan BN, Zhang Y, Lee JH, Cheng R, Bohm C, Ghani M, Reitz C, Reyes-Dumeyer D, Shen Y, Rogaeva E, St George-Hyslop P, Mayeux R. Coding mutations in SORL1 and Alzheimer disease. Ann Neurol. 2015 Feb;77(2):215-27. PubMed.
External Citations
Further Reading
Papers
- Willnow TE, Andersen OM. Sorting receptor SORLA - a trafficking path to avoid Alzheimer disease. J Cell Sci. 2013 Jul 1;126(Pt 13):2751-60. PubMed.
- Scherzer CR, Offe K, Gearing M, Rees HD, Fang G, Heilman CJ, Schaller C, Bujo H, Levey AI, Lah JJ. Loss of apolipoprotein E receptor LR11 in Alzheimer disease. Arch Neurol. 2004 Aug;61(8):1200-5. PubMed.
- Sager KL, Wuu J, Leurgans SE, Rees HD, Gearing M, Mufson EJ, Levey AI, Lah JJ. Neuronal LR11/sorLA expression is reduced in mild cognitive impairment. Ann Neurol. 2007 Dec;62(6):640-7. PubMed.
- Caglayan S, Takagi-Niidome S, Liao F, Carlo AS, Schmidt V, Burgert T, Kitago Y, Füchtbauer EM, Füchtbauer A, Holtzman DM, Takagi J, Willnow TE. Lysosomal sorting of amyloid-β by the SORLA receptor is impaired by a familial Alzheimer's disease mutation. Sci Transl Med. 2014 Feb 12;6(223):223ra20. PubMed.
- Mehmedbasic A, Christensen SK, Nilsson J, Rüetschi U, Gustafsen C, Poulsen AS, Rasmussen RW, Fjorback AN, Larson G, Andersen OM. SorLA complement-type repeat domains protect the amyloid precursor protein against processing. J Biol Chem. 2015 Feb 6;290(6):3359-76. Epub 2014 Dec 18 PubMed.
- Buggia-Prévot V, Thinakaran G. Sorting the role of SORLA in Alzheimer's disease. Sci Transl Med. 2014 Feb 12;6(223):223fs8. PubMed.
Primary Papers
- Kitago Y, Nagae M, Nakata Z, Yagi-Utsumi M, Takagi-Niidome S, Mihara E, Nogi T, Kato K, Takagi J. Structural basis for amyloidogenic peptide recognition by sorLA. Nat Struct Mol Biol. 2015 Mar;22(3):199-206. Epub 2015 Feb 2 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.