New C9ORF72 Antibodies Find Isoforms in Different Cellular Locations
Quick Links
The C9ORF72 gene has been linked to both amyotrophic lateral sclerosis and frontotemporal dementia. Researchers trying to understand the function of the C9ORF72 protein have, until recently, had to make do with fairly poor antibodies. These were unable to distinguish the two natural 54- and 24-kilodalton isoforms, which arise through alternative splicing. In the July 14 Annals of Neurology online, scientists report two new C9ORF72 antibodies. Developed by Shangxi Xiao in the University of Toronto laboratory of Janice Robertson, they distinguish between the short and long isoforms, allowing the scientists to analyze these two versions separately. The antibodies revealed that the short form populates the nuclear membrane of neurons, while its long counterpart remains in the cytoplasm. Further, it looks like the short version might usher another ALS- and FTD-linked protein into the nucleus—TDP-43.
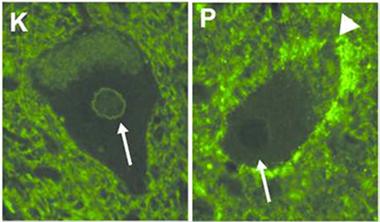
Multiple membranes.
In spinal motor neurons from neurologically healthy people (top), the short form of C9ORF72 attaches to the nuclear membrane, but it localizes to the plasma membrane in tissue from people with ALS (bottom). [Image courtesy of Wiley/Annals of Neurology © American Neurological Association]
While hexanucleotide expansions in C9ORF72 first attracted scientists to this protein, they still have no clear idea what the wild-type protein does (see Sep 2011 news). Commercial antibodies to C9ORF72 were already available in 2011, but they offered poor specificity and sensitivity, said Adrian Waite of the Cardiff University School of Medicine in the United Kingdom. Waite tackled the problem with his own C9ORF72 antibody, which recognizes the amino terminus of the protein. However, it does not distinguish between the two isoforms (Waite et al., 2014). Tania Gendron of the Mayo Clinic in Jacksonville, Florida, said this pan-C9ORF72 antibody is a useful tool, but that isoform-specific antibodies were long overdue. To deliver on those, Xiao took advantage of a terminal lysine unique to the short isoform to generate a synthetic peptide to match. He used this and a peptide found only in the long isoform to immunize rabbits and generate polyclonal antibodies. Now, Xiao and Robertson offer a first glimpse of how these antibodies can help scientists understand C9ORF72. "I think they are going to be a phenomenal resource," Gendron told Alzforum.
On western blots of human cerebellar extracts, Xiao determined that the short form of C9ORF72 may form a dimer. The antibody to the short form recognized two proteins, one being twice the predicted size. The antibody to the large isoform detected a single band of the expected 54 kDa. Xiao found the same pattern in postmortem extracts from controls and from ALS cases—with or without the C9ORF72 expansion gene. “The antibodies look really clean,” Gendron commented.
To see where the two isoforms localize in cells, Xiao turned to immunohistochemistry. In spinal motor neurons from controls and ALS cases, the long C9ORF72 typically appeared in the cytoplasm, offering little clue to its function. In contrast, the short form settled on the nuclear membrane in the majority of control neurons. Interestingly, it was absent from the nucleus in about 60 percent of the neurons in ALS cases. It turned up on the plasma membrane instead (see image above). The pattern was the same whether the ALS was linked to C9ORF72 expansions or not.
Intrigued by the altered localization of the short isoform, Xiao investigated further. In controls, the short form co-localized on the nucleus with Ran-GTPase and importin-β1, which regulate the trafficking of proteins into the nucleus. Like short C9ORF72, these two nuclear import factors were missing from the nuclear membranes of many cells in ALS cases. To check if C9ORF72 directly interacted with these nuclear shuttlers, Xiao used mouse neuroblastoma N2a cultures. When he overexpressed C9ORF72 in those cells, it co-immunoprecipitated with both RAN-GTPase and importin-β1. Thus, the authors suspect C9ORF72 might participate in nuclear import as well.
Other studies have also linked nuclear shuttling to ALS. TDP-43 normally resides in the nucleus but is often found in the cytoplasm of diseased neurons. Scientists have blamed faulty nuclear uptake for this mislocalization (Nishimura et al., 2010; Ward et al., 2014). Might C9ORF72 be involved? Xiao stained human spinal cords for TDP-43 and short C9ORF72. In cells where C9ORF72 had quit the nuclear membrane, TDP-43 was mislocalized to the cytoplasm.
Based on these data, Robertson and Xiao theorize that C9ORF72 abnormalities could cause the short isoform to depart the nuclear membrane, making it impossible for TDP-43 to get into the nucleus. Notably, because the short C9ORF72 vacated the nucleus in sporadic cases of ALS, the authors believe that not just the C9 hexanucleotide expansion but other factors influence C9ORF72 localization and thus TDP-43 import. They plan to look at this in more detail.
Gendron was intrigued by the proposed mechanism, but said she would like to see direct evidence that C9ORF72 participates in nuclear import, for example by knocking down the gene in human cell models.
Waite pointed out that bioinformatics analysis of C9ORF72 suggests it could be a GTP exchange factor, and he speculated it might be on the nuclear membrane to regulate Ran-GPTase (see Jan 2013 news). However, he noted other recent data that might contradict Xiao’s model (see full comment below). For example, one group eliminated neural C9ORF72 in mice and saw no TDP-43 mislocalization (see Jun 2015 news). Robertson pointed out that mice lack the short C9ORF72 isoform, and that their TDP-43 differs from the human version in both sequence and splice variants. She said C9ORF72 knockout mice would not necessarily mimic all aspects of ALS in people.—Amber Dance
References
News Citations
- Corrupt Code: DNA Repeats Are Common Cause for ALS and FTD
- C9ORF72 Function: Is the ALS Protein a Membrane Traffic Cop?
- No C9ORF72, No Problem: Knockout Mouse Neurologically OK
Paper Citations
- Waite AJ, Bäumer D, East S, Neal J, Morris HR, Ansorge O, Blake DJ. Reduced C9orf72 protein levels in frontal cortex of amyotrophic lateral sclerosis and frontotemporal degeneration brain with the C9ORF72 hexanucleotide repeat expansion. Neurobiol Aging. 2014 Jul;35(7):1779.e5-1779.e13. Epub 2014 Jan 17 PubMed.
- Nishimura AL, Zupunski V, Troakes C, Kathe C, Fratta P, Howell M, Gallo JM, Hortobágyi T, Shaw CE, Rogelj B. Nuclear import impairment causes cytoplasmic trans-activation response DNA-binding protein accumulation and is associated with frontotemporal lobar degeneration. Brain. 2010 Jun;133(Pt 6):1763-71. PubMed.
- Ward ME, Taubes A, Chen R, Miller BL, Sephton CF, Gelfand JM, Minami S, Boscardin J, Martens LH, Seeley WW, Yu G, Herz J, Filiano AJ, Arrant AE, Roberson ED, Kraft TW, Farese RV Jr, Green A, Gan L. Early retinal neurodegeneration and impaired Ran-mediated nuclear import of TDP-43 in progranulin-deficient FTLD. J Exp Med. 2014 Sep 22;211(10):1937-45. Epub 2014 Aug 25 PubMed.
External Citations
Further Reading
Papers
- Miller BL. The C9ORF72 mutation brings more answers and more questions. Alzheimers Res Ther. 2013 Feb 18;5(1):7. PubMed.
- Barmada SJ, Skibinski G, Korb E, Rao EJ, Wu JY, Finkbeiner S. Cytoplasmic mislocalization of TDP-43 is toxic to neurons and enhanced by a mutation associated with familial amyotrophic lateral sclerosis. J Neurosci. 2010 Jan 13;30(2):639-49. PubMed.
Primary Papers
- Xiao S, MacNair L, McGoldrick P, McKeever PM, McLean JR, Zhang M, Keith J, Zinman L, Rogaeva E, Robertson J. Isoform-specific antibodies reveal distinct subcellular localizations of C9orf72 in amyotrophic lateral sclerosis. Ann Neurol. 2015 Oct;78(4):568-83. Epub 2015 Aug 29 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Institut Jožef Stefan
In the years since the discovery that the hexanucleotide repeat expansion mutation in C9ORF72 is one of the major causes of ALS and FTLD, we have been presented with three major pathogenesis hypotheses: haploinsufficiency, RNA toxicity, and dipeptide repeat toxicity. Most of the subsequent research has focused on the last two, as they are thought to be more likely root causes of disease. Besides giving insight into disease pathogenesis, this research has made significant contributions to our understanding novel concepts in basic cellular mechanisms, such as RNA aggregation and repeat-associated non-ATG translation.
In this paper, Janice Robertson’s group shifts the attention back to the C9ORF72 protein. They developed two antibodies that distinguish between the long and short isoforms of C9ORF72. The primary characterization of these antibodies reveals some striking ALS-related changes in their subcellular localization as well as possible interactions with two components of nucleocytoplasmic shuttling, a small GTPase RAN (ras-related nuclear protein) and KPNB1 (Importin-b1). The final figure in the paper, where the authors show correlation between RAN/KPNB1, C9ORF72 short form, and TDP-43 aggregation, is fascinating and, if proven by a larger study, will give significant insights into the involvement of nuclear transport in ALS. Importantly, it should also be noted that both RAN and KPNB1 are involved in cell cycle progression so it will be interesting to see if and where the C9ORF72 protein isoforms fit in that process. Five years ago we showed that TDP-43 is transported into the nucleus via the KPNB1 pathway (Nishimura et al., 2010) and recently we showed that loss of TDP-43 also leads to loss of components of nucleocytoplasmic shuttling in human cells, suggesting a feedback regulation of TDP-43 shuttling (Stalekar et al., 2015). One such component is RANBP1 (RAN binding protein 1), which is necessary for facilitating the GTPase function of RAN. Overall, these antibodies present an important new tool, which surely will enable significant advances in characterization of C9ORF72 function as well as understanding its role in disease.
References:
Nishimura AL, Zupunski V, Troakes C, Kathe C, Fratta P, Howell M, Gallo JM, Hortobágyi T, Shaw CE, Rogelj B. Nuclear import impairment causes cytoplasmic trans-activation response DNA-binding protein accumulation and is associated with frontotemporal lobar degeneration. Brain. 2010 Jun;133(Pt 6):1763-71. PubMed.
Štalekar M, Yin X, Rebolj K, Darovic S, Troakes C, Mayr M, Shaw CE, Rogelj B. Proteomic analyses reveal that loss of TDP-43 affects RNA processing and intracellular transport. Neuroscience. 2015 May 7;293:157-70. Epub 2015 Mar 2 PubMed.
Cardiff University
Studies examining the role of the C9ORF72 protein isoforms have been hampered by the lack of commercial C9ORF72 antibodies with high specificity and sensitivity. Currently available reagents are also unable to individually detect the two C9ORF72 isoforms. This study describes the generation and preliminary use of novel isoform-specific C9ORF72 antibodies. The authors developed novel rabbit polyclonal antibodies against synthetic peptides specific to C9ORF72 long and short isoforms. They showed that the antibodies could selectively detect recombinant C9ORF72 isoforms. The antibodies also appear to show increased sensitivity for detection of C9ORF72 isoforms in immunoblotting studies of postmortem tissue samples compared to a Santa Cruz commercial antibody.
Interestingly, the authors show different subcellular localization of the two C9ORF72 isoforms using immunohistochemistry in postmortem tissue, with the short isoform localizing to the nuclear envelope whereas the long isoform shows a diffuse cytoplasmic localization. Furthermore, the levels and subcellular localization of C9ORF72 isoforms are also differentially altered in ALS cases with and without the pathological C9ORF72 hexanucleotide expansion. The authors show that loss of nuclear envelope localization of C9ORF72 short form may correlate with TDP-43 pathology in spinal motor neurons of ALS cases. The study suggests that C9ORF72 could function in nucleocytoplasmic shuttling and that dysfunction of this pathway could be associated with TDP-43 pathology.
They provide evidence that C9ORF72 may interact with the nucleocytoplasmic transport complex components importin-β1 and Ran-GTPase. Mislocalization of these proteins and C9ORF72 short isoform may correlate with TDP-43 pathology in ALS cases with or without pathological C9ORF72 repeat expansions. Evidence of a direct interaction between stably overexpressed C9ORF72 and importin complex components was shown. Further validation of this interaction by immunoprecipitation of endogenous C9ORF72 is required.
The authors use recombinant C9ORF72 isoform expression and the addition of an antibody-blocking peptide for the purposes of showing antibody specificity for immunoblotting and immunohistochemistry. However, further studies should be performed using C9ORF72-deficient cells or tissue to verify specificity of the antibodies.
This study emphasises how the hexanucleotide repeat expansion in C9ORF72 exerts multiple effects in the central nervous system. In addition to the generation of hexanucleotide-derived toxic RNA and dipeptide species there are differential effects on the protein levels and localization of C9ORF72 isoforms. Increasing evidence from animal and cellular models supports gain of function as the main pathological mechanism, whereas haploinsufficiency of C9ORF72 either represents epi-phenomena or could be a disease modifier. Mislocalization of C9ORF72 short form in motor neurons may also be a pathological correlate of ALS with TDP-43 pathology. The finding of C9ORF72 mislocalization in motor neurons from ALS cases without a hexanucleotide expansion and normal localisation in cerebellar Purkinje cells (that have been shown to contain RNA foci and C9RANT pathology) suggests this is not a specific consequence of the hexanucleotide expansion products. Further immunohistochemical studies of tissue with a higher burden of RNA foci and C9RANT pathology, such as frontal cortex and hippocampal tissue, may help address this issue.
Further studies will be required to examine the role of C9ORF72 in nucleocytoplasmic shuttling and to explore whether mislocalization of C9ORF72 short form is a modifier of TDP-43 pathology in ALS or a downstream consequence of TDP-43 dysfunction. Bioinformatic analyses and preliminary functional studies of C9ORF72 suggest that it may be a member of the DENN family of GTP exchange factors (GEFs) and could regulate GTPase activity. Therefore C9ORF72 could regulate nucleocytoplasmic shuttling via GEF activity towards Ran-GTPase. However, a recent study published by Jeroen Pasterkamp and colleagues generated mice with neural-specific ablation C9ORF72 and showed no motor neuron degeneration or pathological hallmarks of ALS such as mislocalization of TDP-43 (Koppers et al., 2015). This would suggest that C9ORF72 deficiency does not affect localization of TDP-43 and that the reported mislocalization of C9ORF72 short form could be a result of dysfunction of TDP-43 or nucleocytoplasmic shuttling. Alterations in nucleocytoplasmic transport could also be a downstream effect of arginine-rich C9RANT products on Ran-GTPase alternative splicing (Kwon et al., 2014).
References:
Koppers M, Blokhuis AM, Westeneng HJ, Terpstra ML, Zundel CA, Vieira de Sá R, Schellevis RD, Waite AJ, Blake DJ, Veldink JH, van den Berg LH, Pasterkamp RJ. C9orf72 ablation in mice does not cause motor neuron degeneration or motor deficits. Ann Neurol. 2015 Sep;78(3):426-38. Epub 2015 Jul 3 PubMed.
Kwon I, Xiang S, Kato M, Wu L, Theodoropoulos P, Wang T, Kim J, Yun J, Xie Y, McKnight SL. Poly-dipeptides encoded by the C9orf72 repeats bind nucleoli, impede RNA biogenesis, and kill cells. Science. 2014 Sep 5;345(6201):1139-45. Epub 2014 Jul 31 PubMed.
Make a Comment
To make a comment you must login or register.