Revised Again: Alzheimer's Diagnostic Criteria Get Another Makeover
Quick Links
Diagnostic criteria for Alzheimer’s disease were revised only five years ago, but the field has learned a lot since then. At the Alzheimer’s Association International Conference, held last month in Amsterdam, Clifford Jack of the Mayo Clinic in Rochester, Minnesota, proposed updates to the current “ATN” system. Like the 2018 criteria, the new ones are the product of a joint National Institute on Aging-Alzheimer’s Association working group Jack leads. The draft criteria take into account recent discoveries about how biomarkers change over time, using them to stage disease. They retain amyloid and tau for diagnosis and staging but relegate the “N” neurodegeneration marker to second-tier status. They also advance the previous research diagnostic criteria toward clinical use by selecting the best-validated biomarkers.
- Revised ATN draft drops the N for diagnosis and staging of Alzheimer’s.
- Either fluid or PET A and T biomarkers can be used. They can't be mixed.
- N becomes a second-tier marker, along with I (inflammation) and co-pathologies.
With two disease-modifying AD treatments already in the clinic, and a third expected, accurate diagnosis has become an urgent need.
The criteria debuted to a packed hall at AAIC. Audience members peppered a panel of working group members with questions for nearly an hour, running the session well into dinnertime. While some aspects of the new criteria raised questions, the overall reception was positive.
“This is great progress, absolutely the direction we need to go,” Gil Rabinovici of the University of California, San Francisco, told the panel. Randall Bateman of Washington University in St. Louis was equally enthusiastic. “This [system] matches the biology so well. You’ve nailed it,” he said in Amsterdam. Bateman acted as a reviewer for the working group, but was not part of developing the criteria.

From ATN To ATNIVS. In the proposed new scheme—which is currently a draft meant to solicit input from the ADRD research community—A and T are the core biomarkers for diagnosis and staging. The draft scheme also recognizes an expanded suite of additional markers that detect non-specific disease responses and co-pathologies. [Courtesy of NIA-AA working group.]
Evolving Disease Conceptions
In 2018, the NIA-AA committee established the first biological definition of Alzheimer’s disease, basing diagnosis on the presence of amyloid plaques (A) and tau tangles (T), rather than symptoms. The criteria also made use of biomarkers of neurodegeneration (N) to help stage disease. The system distinguished clinical stages from 1 to 6, with 1 being biomarker change only, 2, subtle cognitive change, 3, mild cognitive impairment, and 4-6, mild, moderate, and severe dementia (Apr 2018 news; Nov 2018 conference news). The criteria were intended to be a work in progress, and to incorporate new data once it was available.
In Amsterdam, researchers said that moment has come. Jack noted several major changes in the field in the last five years. Besides FDA approval of disease-modifying therapies, the advent of plasma biomarkers plus the recognition that fluid and imaging biomarkers are not interchangeable required updating the standards, he said.
To do this, the NIA-AA convened a 22-person working group. In addition to Jack, academic researchers comprised Thomas Beach of Banner Sun Health Research Institute in Sun City, Arizona; Oskar Hansson of Lund University, Sweden; William Jagust of the University of California, Berkeley; Eric McDade of Washington University in St. Louis; Ozioma Okonkwo of the University of Wisconsin, Madison; Luca Pani of the University of Modena and Reggio Emilia, Italy; Michael Rafii of the University of Southern California, Los Angeles; Philip Scheltens, recently retired from VU University, Amsterdam; Reisa Sperling of Brigham and Women’s Hospital, Boston; and Charlotte Teunissen of Amsterdam UMC.
From pharma, members comprised Scott Andrews at Takeda Pharmaceuticals, Ana Graf at Novartis, Carole Ho at Denali Therapeutics, José Luis Molinuevo at Lundbeck, and Eric Siemers at Acumen Pharmaceuticals. The committee included Eliezer Masliah and Laurie Ryan from the NIA, Teresa Buracchio and Billy Dunn from the Food and Drug Administration, and Maria Carrillo and Heather Snyder from the Alzheimer’s Association. Dunn left the FDA in February; he serves on the board of Prothena. The group has been working on the draft criteria for a year, and is now soliciting outside feedback from the field, Jack said.
Diagnosis by Amyloid and Tau Only What are the new criteria? For starters, the diagnostic scheme ditches the N category, relying only on A and T, which are considered the core biomarkers of the disease. This change was made because amyloid and tau pathology are more specific for AD than are neurodegeneration markers, Jack said. Moreover, the new approach allows clinicians to use either fluid or imaging biomarkers for diagnosis, with equal validity. Researchers hope this will make AD diagnosis feasible even in parts of the world where brain imaging is unavailable.
The committee specified the biomarkers they believe to be sufficiently validated for clinical use (see table above). For A, these are the Aβ42/40 ratio in cerebrospinal fluid or plasma, and amyloid PET imaging. For T, these are p-tau181 or p-tau217 in CSF or plasma, and tau PET imaging. Other biomarkers, such as p-tau231 and p-tau205, may reach this level later, but for now should be restricted to research use.
Bateman questioned the use of p-tau181 and p-tau217 as tau markers, noting they are more closely tied to plaques than tangles (Dec 2019 conference news; Mar 2020 news). P-tau205, MTBR-tau243, or even total tau, would make better markers for tangles, he suggested (Dec 2022 conference news; Part 3 of this series). Hansson explained that the committee conceived of T as denoting a change in tau metabolism, not specifically the presence of tangles, but agreed the inclusion of p-tau181 and p-tau217 in the T category might cause people to misinterpret them as tangle markers.
The draft criteria are still being refined and may change, Jack said. Indeed, part of the purpose in presenting the draft proposals at AAIC was to invite comment from the global community of ADRD clinician-researchers.
Importantly, the draft criteria currently do not specify particular assays or cutoffs to be used for each biomarker. This should be up to clinicians, and the changing state of the field. However, the criteria do recommend that instead of a single cut point, clinicians recognize an “indeterminate zone” for each biomarker, where a person may or may not have the disease. Results in this zone would trigger further testing.
A point of controversy in Amsterdam was that, in the new scheme, AD can be diagnosed by the presence of either A or T alone. This elicited audience pushback. After all, some people with plaques don’t go on to develop tangles. Some argued that amyloid plaques should be considered a risk factor for AD, rather than diagnostic of it.
The panel disagreed. Amyloidosis is the first event in AD, Jack said. In other words, people who have plaques in their brain already have the disease. They are at risk for its symptoms. Sperling concurred. “Not everyone with amyloid will develop symptoms, but we take [its presence] seriously, and we want to treat it,” she said in Amsterdam. McDade noted that the proposed NIA-AA criteria do not advocate screening cognitively healthy people for plaques. Thus, everyone diagnosed as A+ is likely to have come to a memory clinic with cognitive concerns.
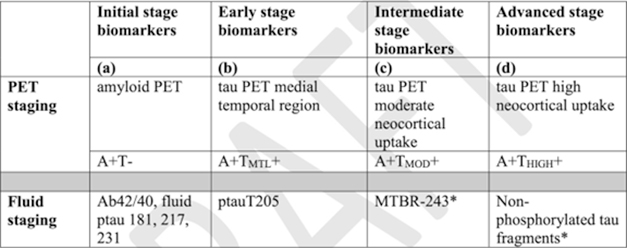
Parallel Tracks. The proposed criteria, shown here in draft form dated July 15, allow clinicians to stage disease by either PET or fluid markers; alas, the stages denoted by each are not equivalent, and the modalities cannot be mixed. Late-stage fluid biomarkers are less well-established than the others, with the starred markers meant to be used only in research settings at this time. [Courtesy of NIA-AA working group.]
Disease Staging Via Parallel Tracks—Fluid or Imaging
In addition to diagnosing disease, A and T markers are able stage it, Jack told the audience. This is possible because scientists have recently associated specific A and T markers with distinct disease stages. The new framework specifies four biomarker stages: initial, early, intermediate, and advanced. These are denoted by letters A through D, to distinguish the biomarker stages from the clinical stages carried over from 2018, which are numerical.
The framework allows for two separate diagnostic paths, one using PET imaging and the other fluid biomarkers. However, Jack emphasized that stages determined by fluid markers or imaging are not equivalent to each other. For example, although the early and intermediate fluid biomarkers p-tau205 and MTBR-tau243 are correlated with tau tangles, researchers have not yet determined how they relate to the location and severity of those tangles as seen by PET. Clinicians will need to stick to one modality for a given patient, or when comparing data between patients.
Within the PET staging pathway, initial disease is diagnosed by global amyloid PET positivity, early disease by tau tangles in the temporal cortex, intermediate disease by a low level of tangles in the neocortex, and advanced disease by high neocortical tangles. For tau PET, both the location and magnitude of uptake matters, Jack noted.
How well does this PET staging work? In Amsterdam, Jack showed two different tests of the method. In one, researchers at ADNI and the Australian Imaging, Biomarkers, and Lifestyle study evaluated 1,045 participants staged in the new way. They found that later stages correlated with a greater prevalence of cognitive impairment and dementia, as expected (Villemagne et al., 2023). In the other, researchers led by Hansson and Rik Ossenkoppele at Lund analyzed progression to MCI and dementia over six years among 1,325 people staged by the new method. They found increasingly greater hazard ratios for clinical progression at more advanced PET stages. For example, compared with cognitively healthy people who were A-T-, those who were A+T- were twice as likely to progress to MCI, A+TMTL+ were 15 times as likely, and A+TNEO+ were 19 times as likely (Ossenkoppele et al., 2022).
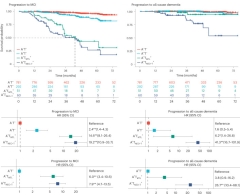
Better Predict Progression. People at more advanced PET stages of disease (dark blue, green) develop cognitive impairment (left) and dementia (right) faster than do those at early PET stages (red, light blue). [Courtesy of Ossenkoppele et al., 2022.]
In the fluid biomarker pathway, initial disease is diagnosed by an abnormal CSF Aβ42/40 ratio, or elevated p-tau181, p-tau217, or p-tau231. Early disease is marked by elevated p-tau205, intermediate by MTBR-tau-243, and advanced by a new marker, non-phosphorylated tau fragments in the CSF (Montoliu-Gaya et al., 2023). Jack noted that fluid biomarker staging is more tentative than the PET staging scheme, and needs additional validation.
More Markers: Nonspecific Reactions and Co-Pathologies
While amyloid and tangles are the core biomarkers for AD diagnosis, the new draft criteria recognize that nonspecific reactions contribute. Take neurodegeneration (N) and inflammation (I). They are nonspecific because they occur in many neurodegenerative diseases. Validated N biomarkers include NfL, volumetric MRI, and FDG PET; I has only GFAP so far. Many more biomarkers, such as fluid neurogranin and synaptic PET imaging for N, and fluid YKL-40 and sTREM2 for I, are in research use and may become fit for clinical use.
Finally, the criteria specify markers of co-pathology. Chief among these are vascular damage (V) and α-synuclein (S). For the former, markers include MRI of white-matter hyperintensities, expanded perivascular spaces, and infarctions. For the latter, α-synuclein seed amplification assays of CSF samples are up and coming (Apr 2023 conference news; Part 9 of this series). As yet, there are no markers for TDP-43 deposits, another common co-pathology.
In some cases, the full ATNIVS suite of biomarkers will show mismatches, Jack said. He presented an example of a man who had dementia and was positive for A and N biomarkers, but negative for T. Although the man had Alzheimer’s disease, that was not the cause of his dementia, Jack said. Instead, the culprit was advanced limbic predominant age-related TDP-43 encephalopathy (LATE), which often co-occurs with AD pathology and can confound diagnosis (May 2019 news; Mar 2022 conference news).
Several audience members asked how clinicians will know whether a particular pathology in a given patient is driving that person’s clinical syndrome. The answer? Physicians must interpret biomarkers in the context of each patient’s medical history. For example, poor kidney function, obesity, and certain medications can hike a person’s level of AD biomarkers. Head injuries are known to spike p-tau231. All of that will have to be taken into account. “That’s why us clinicians will still have jobs,” Sperling quipped.
The new staging scheme represents the natural progression of disease, in the absence of treatment. An Alzheimer’s patient who becomes amyloid-negative after a course of immunotherapy treatment still has the disease, Jack said, in answer to an audience question. “Disease-modifying treatments may alter the relationships between biomarkers, but the disease does not disappear,” he added.
How Do Clinical Stages Fit In?
For clinical staging, the committee largely kept the method from 2018. They made only one change, adding a stage zero. It is for people who inherit deterministic AD mutations but do not yet have any biomarker or clinical changes.
How to integrate biomarker and clinical staging? The classical progression would be for a person to move from stage 1A (no symptoms, initial biomarkers) to 2B (subtle symptoms, early biomarkers), 3C (MCI, intermediate biomarkers), and finally 4-6D (dementia, advanced biomarkers). However, an individual person's progression might be different, Jack said. Some people will fare worse clinically than would be expected based on their A and T biomarkers. This could indicate the presence of co-pathologies, which can bring on neurodegeneration and cognitive symptoms earlier than otherwise expected. Others will fare better than expected, perhaps due to resilience factors. “Clinical impairment does not scale in lockstep with biomarkers,” Jack said.
Jack acknowledged that the new system is more complicated than the old. This is also true in other diseases such as cancer, where diagnostic and staging schemes become more elaborate as scientists learn more. The greater level of detail will help better tailor diagnosis to individuals, leading to more personalized medicine, he predicted. Jack also believes that the ability to target treatments to particular disease stages will become more sophisticated. “The era of disease-modifying treatments for AD has just begun,” he noted.
The authors invite feedback here. —Madolyn Bowman Rogers
References
News Citations
- New Definition of Alzheimer’s Hinges on Biology, Not Symptoms
- How Does The NIA-AA Framework Measure Up Against Real Data?
- Blood Tests of Phospho-Tau, Aβ42, Track With Brain Amyloid
- Different CSF Phospho-Taus Match Distinct Changes in Brain Pathology
- New Data Bolsters MTBR-Tau243 as Fluid Marker for Tangles
- CSF MTBR-tau-243 Tracks Tangles, Plummets in Response to Antibody
- Synuclein Assay Passes the Sniff Test—What of Other Seeds?
- Finally, a Diagnostic Marker for Lewy Body Disease?
- Introducing LATE—A Common TDP-43 Proteinopathy that Strikes After 80
- Does LATE Subvert Alzheimer's Trials? Biomarkers, Please!
Paper Citations
- Villemagne VL, Leuzy A, Bohorquez SS, Bullich S, Shimada H, Rowe CC, Bourgeat P, Lopresti B, Huang K, Krishnadas N, Fripp J, Takado Y, Gogola A, Minhas D, Weimer R, Higuchi M, Stephens A, Hansson O, Doré V, Alzheimer's Disease Neuroimaging Initiative and the AIBL research group. CenTauR: Toward a universal scale and masks for standardizing tau imaging studies. Alzheimers Dement (Amst). 2023;15(3):e12454. Epub 2023 Jul 7 PubMed.
- Ossenkoppele R, Pichet Binette A, Groot C, Smith R, Strandberg O, Palmqvist S, Stomrud E, Tideman P, Ohlsson T, Jögi J, Johnson K, Sperling R, Dore V, Masters CL, Rowe C, Visser D, van Berckel BN, van der Flier WM, Baker S, Jagust WJ, Wiste HJ, Petersen RC, Jack CR Jr, Hansson O. Amyloid and tau PET-positive cognitively unimpaired individuals are at high risk for future cognitive decline. Nat Med. 2022 Nov;28(11):2381-2387. Epub 2022 Nov 10 PubMed.
- Montoliu-Gaya L, Benedet AL, Tissot C, Vrillon A, Ashton NJ, Brum WS, Lantero-Rodriguez J, Stevenson J, Nilsson J, Sauer M, Rahmouni N, Brinkmalm G, Lussier FZ, Pascoal TA, Skoog I, Kern S, Zetterberg H, Paquet C, Gobom J, Rosa-Neto P, Blennow K. Mass spectrometric simultaneous quantification of tau species in plasma shows differential associations with amyloid and tau pathologies. Nat Aging. 2023 Jun;3(6):661-669. Epub 2023 Apr 27 PubMed.
External Citations
Further Reading
Annotate
To make an annotation you must Login or Register.

Comments
MGH
University of Southern Denmark
UCLA School of Medicine
Hospital of the University of Pennsylvania
Revision of Alzheimer’s Diagnostic Criteria: Double Negatives Won’t Do the Job
The diagnostic criteria for Alzheimer’s disease (AD) have undergone a number of changes since 1984 (Knopman et al., 2019). Originally, they were based mainly on clinical assessment, and then from 1997 onward primarily on postmortem neuropathological findings, namely amyloid plaques and neurofibrillary tangles. In 2011 the U.S. National Institute on Aging and Alzheimer’s Association (NIA-AA) for research purposes endorsed a diagnosis of preclinical AD based on the presence of positive AD biomarkers in the cerebrospinal fluid (CSF) or according to cerebral amyloid-PET imaging, whereas cognitive impairment was left out as a criterion (Sperling et al., 2011).
A series of authors created in 2018 a NIA-AA Research Framework including the “ATN” classification (Jack et al., 2018) based on an “unbiased descriptive classification scheme for Alzheimer disease biomarkers” proposed by themselves in 2016 (Jack et al., 2016). The “A/T/(N)” classification, as it was written in 2018, put crucial emphasis on “A,” biomarkers of Aβ plaques, and “T,” biomarkers of tau, whereas biomarkers of neurodegeneration “(N),” including FDG-PET hypometabolism and atrophy on MRI, were in parenthesis, indicating a lesser diagnostic role (Jack et al., 2018).
The latest proposed revision is not only devoid of clinical assessment, it also drops the “N” for diagnosis and staging of Alzheimer's. It relies solely on biomarkers, the pathobiological role of which has never been proven (Kepe et al., 2013; Høilund-Carlsen et al., 2022; Høilund-Carlsen et al., 2023). The proposers wish to endorse it not only for research but also for clinical practice. In the case of the clinical trials, there seems to be a push to define the disease as the presence of surrogate markers (A and T) alone.
It is generally agreed that AD is a form of cognitive decline caused by neurodegeneration, but neither of these characteristics are tested for or given diagnostic value in the revised ATN system, which the authors now want clinicians to use "by selecting the best-validated biomarkers,” none of which have been properly validated. The lack of correlation between cognitive status and the biomarkers in question was highlighted some 25 years ago, as was the lack of a causal relationship between amyloid-beta and AD (Robakis and Pangalos, 1994; Neve and Robakis, 1998), two serious discrepancies that no one has since been able to explain.
If one disregards “T” in order to simplify the issue, the proposed version has, when considering also clinical assessment and imaging, the following eight possible outcomes, where + and – mean presence and absence of amyloid (A), clinical symptoms (S) and imaging abnormalities (I):
According to the revised version, outcomes 1-4 and only these combinations are synonymous with the diagnosis of AD. However, how can a responsible physician accept that outcomes 5-7 have nothing to do with AD? Not only that, how would you convince a physician that outcome 3 (A+/S-/I+) and particularly outcome 4 (A+/S-/I-), where there is evidence of amyloid by PET or fluid biomarkers, but where the patient is cognitively normal with a normal MRI and/or FDG-PET, is actually suffering from AD? These would be jarring diagnoses, but apparently not to the ears of the proposers.
To us, the combination of S+ and I+ is the only one that makes sense, i.e., only outcomes 1 and 5 are reasonably certain to indicate AD, whereas the other six outcomes cannot be described as either early or definite AD. Of these, outcomes 2-4 are, according to the proposers, candidates for passive immunotherapy, even though those with outcomes 2 and 3 most likely do not have AD, while those with outcome 4 definitely do not suffer from that.
The double-negatives analogy: How can the absence of symptoms and of reduced brain size and cerebral function “erase” themselves in the making of the AD diagnosis? When referring to the “Uncle Rule” used in the Nordic countries, is that compelling enough for you to advise your uncle (or another family member or yourself) to receive immunotherapy in case of “amyloid positivity”—even in the absence of symptoms and imaging abnormalities? To us, the answer is simple: No! What should the Alzheimer's Association and the relatives say—not to mention the patients, who unfortunately can rarely speak for themselves?
There is no hook in the ceiling to hang the definition on, i.e., an infallible reference to refer to and test against. We propose a definition based on clinical assessment and in vivo evidence of increased neurodegeneration that most clinicians will be able to relate to and use in their daily work (Høilund-Carlsen et al., 2023), supplemented by biomarkers with the proviso that the biomarkers are acknowledged to be surrogates that have not yet been shown to be causal despite a 20-year effort to do so.
References:
Knopman DS, Petersen RC, Jack CR Jr. A brief history of "Alzheimer disease": Multiple meanings separated by a common name. Neurology. 2019 May 28;92(22):1053-1059. Epub 2019 Apr 26 PubMed.
Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR Jr, Kaye J, Montine TJ, Park DC, Reiman EM, Rowe CC, Siemers E, Stern Y, Yaffe K, Carrillo MC, Thies B, Morrison-Bogorad M, Wagster MV, Phelps CH. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011 May;7(3):280-92. Epub 2011 Apr 21 PubMed.
Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, Holtzman DM, Jagust W, Jessen F, Karlawish J, Liu E, Molinuevo JL, Montine T, Phelps C, Rankin KP, Rowe CC, Scheltens P, Siemers E, Snyder HM, Sperling R, Contributors. NIA-AA Research Framework: Toward a biological definition of Alzheimer's disease. Alzheimers Dement. 2018 Apr;14(4):535-562. PubMed.
Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Feldman HH, Frisoni GB, Hampel H, Jagust WJ, Johnson KA, Knopman DS, Petersen RC, Scheltens P, Sperling RA, Dubois B. A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology. 2016 Aug 2;87(5):539-47. Epub 2016 Jul 1 PubMed.
Kepe V, Moghbel MC, Långström B, Zaidi H, Vinters HV, Huang SC, Satyamurthy N, Doudet D, Mishani E, Cohen RM, Høilund-Carlsen PF, Alavi A, Barrio JR. Amyloid-β Positron Emission Tomography Imaging Probes: A Critical Review. J Alzheimers Dis. 2013 May 6; PubMed.
Høilund-Carlsen PF, Revheim ME, Alavi A, Satyamurthy N, Barrio JR. Amyloid PET: A Questionable Single Primary Surrogate Efficacy Measure on Alzheimer Immunotherapy Trials. J Alzheimers Dis. 2022;90(4):1395-1399. PubMed.
Høilund-Carlsen PF, Revheim ME, Costa T, Kepp KP, Castellani RJ, Perry G, Alavi A, Barrio JR. FDG-PET versus Amyloid-PET Imaging for Diagnosis and Response Evaluation in Alzheimer's Disease: Benefits and Pitfalls. Diagnostics (Basel). 2023 Jul 3;13(13) PubMed.
Robakis NK, Pangalos MN. Involvement of amyloid as a central step in the development of Alzheimer's disease. Neurobiol Aging. 1994;15 Suppl 2:S127-9. PubMed.
Neve RL, Robakis NK. Alzheimer's disease: a re-examination of the amyloid hypothesis. Trends Neurosci. 1998 Jan;21(1):15-9. PubMed.
Make a Comment
To make a comment you must login or register.